Introduction
The United States remains in the midst of an overdose crisis with approximately 105,000 drug overdose deaths in 2023, including nearly 80,000 involving opioids (Centers for Disease Control and Prevention 2025). Orthopedic surgery represents the largest source of opioid prescriptions among surgical specialties, accounting for >40% of opioid prescriptions from US surgeons dispensed between 2016 and 2022 (Zhang et al. 2023). Many studies focus on opioid death rates, but opioid-related adverse effects also carry significant cost and morbidity (Benson et al. 2025).
Despite the high volume of opioid prescribing, there is no evidence-based guideline for the number of opioids prescribed following total joint arthroplasty, leaving significant variation in practice (Benson et al. 2025). Emerging evidence has shown that multi-modal and non-opioid strategies including nonsteroidal anti-inflammatory drugs (NSAIDs) and gabapentinoids can reduce opioid use and have modest protective effects on the postoperative period (Syed et al. 2025). However, there remains a paucity of alternative options for pain control following total joint arthroplasty, highlighting the need for further investigation.
As a response to this gap, suzetrigine (Journavx™) has emerged as a potential non-opioid pain management strategy in this surgical setting. Suzetrigine is a non-opioid orally administered medication approved by the FDA that targets voltage-gated sodium channels, specifically Nav1.8, in the peripheral nervous system. It works by blocking afferent pain signals before they reach the brain (Vertex Pharmaceuticals, JOURNAVX clinical trials webpage). To the author’s knowledge, no reports have been made describing the clinical use of suzetrigine in postoperative pain management following total hip and knee arthroplasty. Anecdotally, there have been reports of significant opioid reduction while others report little benefit.
This study investigates whether starting suzetrigine 2 hours prior to surgery, and continuing for 2 weeks postoperatively, can help patients avoid opioid use in the postoperative period when added to a robust second generation multimodal protocol. We hypothesize that suzetrigine can be a useful adjunct in the pursuit of opioid-free recovery after total hip arthroplasty (THA) or total knee arthroplasty (TKA).
Methods
Suzetrigine was FDA approved for the treatment of acute pain in January 2025. The senior author began using suzetrigine in April 2025 in an intermittent fashion. Based on early positive results, the senior author stopped routinely prescribing an opioid as part of the standard post-operative medications, replacing it with suzetrigine beginning June 16, 2025. A retrospective chart review was performed on all patients that underwent THA or TKA from June 16, 2025 to October 31, 2025. All total joint procedures were performed by a single surgeon. A total of 236 consecutive patients were included. One hundred nineteen of these patients had a TKA while 117 had a THA. Patients were identified using Current Procedural Terminology (CPT) codes 27447 and 27130. All patients were prescribed suzetrigine 100mg 2 hours pre-operatively and 50mg every 12 hours post-operatively for 2 weeks. This duration was selected based on the current indication of suzetrigine for the treatment of acute pain for up to 2 weeks and to align with the expected postoperative pain course following surgery. Patient demographics were recorded including age, sex, BMI, and ASA score. General anesthesia was utilized for all patients. Past medical history was also noted for depression, anxiety, antiseizure medications and hyperlipidemia. All patients underwent THA via an anterior approach. Any opioid use prior to the respective operation, or in the postoperative period, was noted and verified through the New York State Internet System for Tracking Over-Prescribing (NYSISTOP) database using a HIPAA-secure portal. Any patient that had a prescription for an opioid in the 90 days prior to surgery was placed in the pre-op opioid user group. All other patients were placed in the opioid naive group. This review was performed to objectively determine preoperative opioid use, and to identify any postoperative opioid prescriptions, regardless of whether they were prescribed by the authors’ office or an external provider.
All patients were treated with a robust, second generation multi modal protocol. This included standardized education and expectation setting preoperatively, preoperative cooling of operative site, preop NSAID and acetaminophen, and dexamethasone 20mg IV on the day of surgery.
Postoperatively, all patients had a low, daily maximum step count advanced weekly, extended length tranexamic acid at 1300mg two times a day in patients with a GFR >45 for seven days and 1300 mg once a day for patients with a GFR >45 for seven days, post-op oral NSAIDs for 30 days unless contra-indicated (e.g. anticoagulation other than aspirin, gastric bypass), prednisone 5mg PO daily for 3 weeks, and a specific, non-aggressive, at-home therapy protocol (Wickline et al. 2023). Hourly icing while awake for the first two weeks was also instituted. Patients were instructed to contact the emergency arthroplasty line for uncontrolled postoperative pain, through which opioid prescriptions could be provided within 24 hours as clinically indicated.
Statistical Product and Service Solutions (SPSS) was used for all statistical analyses. Continuous variables, including age, body mass index (BMI), and American Society of Anesthesiologists (ASA) score were summarized using means and standard deviations. Comparisons between opioid-free patients and those who required postoperative opioids were performed using independent-samples t-tests. Categorical variables, including sex, type of procedure (TKA vs THA), depression, anxiety, hyperlipidemia, preoperative opioid use, and postoperative opioid use status, were summarized as frequencies and percentages. Associations between categorical variables and postoperative opioid use were evaluated using Chi-square tests. For the association between preoperative opioid use and postoperative opioid requirement, odds ratios (ORs) with 95% confidence intervals (CIs) were calculated to quantify the strength of association. Statistical significance was defined as p < 0.05.
Results
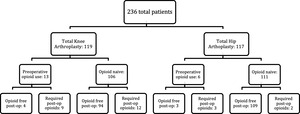
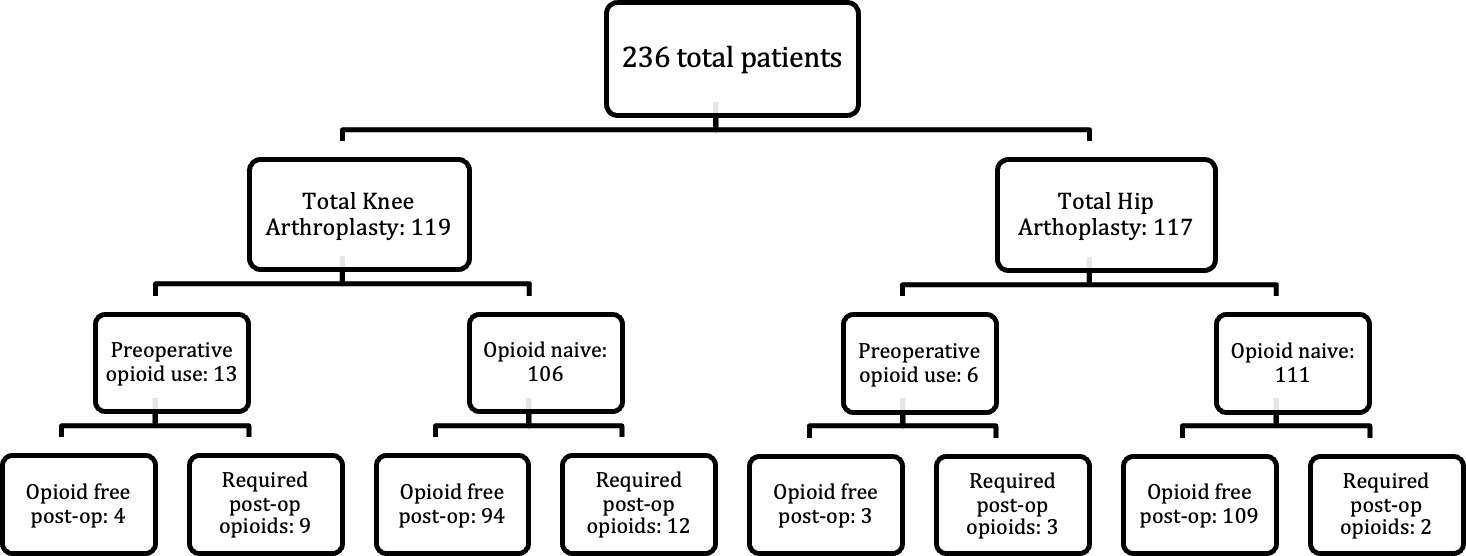
A total of 236 patients undergoing total hip or total knee arthroplasty were included in the study. The mean age of the cohort was 68.5 ± 8.1 years, and 139 patients (58.9%) were female. The mean body mass index (BMI) was 30.7 ± 5.1. 119 patients (50.4%) had a total knee arthroplasty and 117 patients (49.6) had a total hip arthroplasty. Preoperative opioid use within 90 days of surgery was identified in 19 patients (8.1%). Postoperatively, 210 patients (89.0%) never required an opioid prescription, while 26 patients (11.0%) were prescribed opioids within 42 days of surgery. Patient opioid use stratified by procedure type and preoperative along with postoperative opioid status is shown in Figure 1.
Among the 26 patients who required opioid use, tramadol was the most commonly prescribed medication (17 patients, 65.4%), followed by oxycodone (4 patients, 15.4%), and hydrocodone (4 patients, 15.4%). One patient received tramadol–acetaminophen combination formulation (3.8%). The number of pills prescribed ranged from 6 to 150 in the postoperative period with a mean of 42.4 pills per patient in the 42 day postoperative period. 13 out of the 26 patients (50%) were preoperative opioid users with 69% (9/13) of these patients receiving external prescriptions from other providers. Prescriptions in this analysis included those issued by the treating surgeon at the study institution as well as those prescribed by outside providers during the postoperative follow-up period (Table 1).
In the overall study cohort (n = 236), comorbid conditions included depression in 26 patients (11.0%), anxiety in 11 patients (4.7%), and hyperlipidemia in 124 patients (52.5%). All but one patient were classified as ASA II or III.
When comparing opioid-free patients with those who required postoperative opioids, there were no statistically significant differences in age, sex, body mass index, depression, anxiety, hyperlipidemia, or ASA class. Although a higher proportion of patients who required postoperative opioids had an ASA class III compared with opioid-free patients (69.2% vs 57.1%), this difference was not statistically significant (p = 0.238). Procedure type was significantly associated with postoperative opioid use, with patients undergoing TKA more likely to require postoperative opioids than those undergoing THA (80.8% vs 19.2%, p = 0.001).
To further characterize the association between preoperative opioid use and postoperative opioid requirements, outcomes were stratified by procedure type (Tables 4A and 4B). Among opioid-naive patients undergoing TKA, 94 of 106 patients (88.7%) remained opioid-free postoperatively, compared with 109 of 111 patients (98.2%) undergoing THA. In patients with preoperative opioid exposure, some patients were able to achieve opioid free recovery.
Specifically, 4 of 13 TKA patients (30.8%) and 3 of 6 THA patients (50.0%) with preoperative opioid use were opioid-free following surgery (p < 0.001 for both comparisons).
In multivariable analysis, preoperative opioid exposure was independently associated with an increased likelihood of postoperative opioid use despite suzetrigine, with an odds ratio of 24.9 (95% CI, 8.5–73.1; p < 0.001) (Table 5).
Discussion
The opioid epidemic is an undeniable crisis, and an effort to diminish reliance and usage is the responsibility of all providers, including total joint arthroplasty surgeons. To date, consistent opioid-free recovery in large series has not been widely demonstrated. Prior work by the same surgeon demonstrated low postoperative opioid requirements following total knee and total hip arthroplasty using a multimodal protocol without suzetrigine (Stevenson and Wickline 2020). In contrast, the present study demonstrates a shift toward opioid-free recovery, with 82% (98/119) of TKA patients and 96% (112/117) of THA patients not requiring a postoperative opioid prescription.
Previous studies have documented a steady and significant increase in opioid use following total joint replacement, with morphine milligram equivalents rising annually across multiple institutions (Mahmoud et al. 2024). It has been estimated that 8.2% of patients undergoing total knee arthroplasty (TKA) and approximately 4.3% undergoing total hip arthroplasty (THA) develop persistent opioid use at 6 months (Goesling et al. 2016). With annual procedural volumes projected to approach 2.9 million TKAs and nearly 2.0 million THAs by 2060, these rates could correspond to nearly 240,000 new persistent opioid users following TKA and more than 85,000 following THA annually, representing over 320,000 potential new persistent opioid users each year (Shichman et al. 2023).
Understanding current opioid administration patterns is essential for developing safer postoperative pain management strategies and reducing inappropriate or unnecessary prescribing. Decreasing opioid prescriptions may lower the risk of dependence and mitigate adverse effects such as nausea, constipation, and respiratory depression. More cautious prescribing may also reduce medication overutilization, potentially decreasing complications and hospital readmissions while improving overall healthcare efficiency.
A 2024 meta-analysis suggests that typical postoperative requirements after total knee arthroplasty range from approximately 50 to 104 opioid tablets (Dawson et al. 2024).Furthermore, Bridges et al. demonstrated that although surgeons believed they were prescribing a total of 30 oxycodone tablets postoperatively, actual total pills prescribed averaged closer to 110 tablets for total hip and knee arthroplasty (Bridges et al. 2025). Notably, Stevenson et al. and Wickline et al. reported that 86% of total knee arthroplasty patients and 97% of total hip arthroplasty patients required 10 tablets or fewer, highlighting the substantial national differences in prescribing practices.(Stevenson and Wickline 2020 and Wickline et al. 2020) .
Given the procedural volume reported by Jones et al., with approximately 1 million primary total knee arthroplasties and 600,000 primary total hip arthroplasties performed annually, prescribing patterns at 110 oxycodone tablets per procedure could result in approximately 176 million tablets entering communities each year (Jones et al. 2025). Adoption of the reduced prescribing
protocols described in 2020 studies would decrease this estimate to approximately 16 million tablets. Based on the findings of the present study, implementation of optimized opioid-sparing pathways could further reduce this number to roughly 5.4 million tablets annually, representing an approximate 97% reduction in potential community opioid exposure.
Implementation of a second generation analgesic protocol now incorporating suzetrigine was associated with a high rate of opioid-free recovery following total joint arthroplasty. These findings should be interpreted in the context of a comprehensive multimodal pathway, and the independent effect of suzetrigine cannot be isolated. While these findings are encouraging for postoperative arthroplasty pain control, there are important limitations to acknowledge. The study design is retrospective and reflects the experience of a single surgeon. Additionally, suzetrigine was added to a protocol that already yielded low opioid usage. Surgeons should not expect that the addition of suzetrigine alone, to older, first generation multimodal protocols will allow their patients to suddenly be opioid-free. Future multi-center prospective studies comparing opioid-free protocols with standard total joint arthroplasty regimens are recommended.