Introduction
Hip fractures are among the most common fragility fractures in patients with osteoporosis (Pasco et al. 2006), and their impact accounts for >40% of the global clinical burden of osteoporosis (Johnell and Kanis 2006) and >70% of the overall costs (Burge et al. 2007). History of hip fracture is a strong predictor of future hip fractures (Colón-Emeric et al. 2003; Gehlbach et al. 2012; Schemitsch et al. 2022). Approximately 1 in 4 patients die within a year of hip fracture (Abrahamsen et al. 2009; Bentler et al. 2009; Cooper 1997; Leibson et al. 2002), and a second hip fracture carries an additional 25% one-year mortality risk (Schemitsch et al. 2022). Hip fractures are also associated with loss of independence and substantial reductions in functional status and quality of life (Bentler et al. 2009; Cooper 1997; Tajeu et al. 2014; Black et al. 1996). A study of US Medicare patients showed that a hip fracture increased the likelihood of requiring long-term nursing facility care by 4-fold, and many patients who become debilitated after hip fracture also experience financial destitution within a year (Tajeu et al. 2014).
A limited number of systemic osteoporosis medications reduced hip fracture risk in pivotal trials, including alendronate (Black et al. 1996), risedronate (McClung et al. 2001), zoledronic acid (Black et al. 2007), denosumab (Cummings et al. 2009) and romosozumab (Saag et al. 2017). These agents were shown to reduce hip fracture risk by 30-50% and also reduce the risk of non-hip fractures, making them important treatment options for treating osteoporosis. However, none of these agents reduced hip fracture risk in less than one year, and many patients fail to adhere to these agents beyond one year (Ferrari et al. 2016). Moreover, no osteoporosis pharmacotherapy has been proven to reduce the risk of a second hip fracture. Thus, there remains an unmet need for treatment approaches that rapidly strengthen hips to reduce the risk of hip fracture, particularly in patients at high risk of a second hip fracture.
A local osteo-enhancement procedure (LOEP) has been developed to address bone loss, including bone loss at the hip and spine (Howe et al. 2020; Stroncek et al. 2019; Trost et al. 2020). LOEP entails local implantation of AGN1 into surgically prepared bone voids. AGN1 is an osteoconductive, triphasic material consisting of calcium sulfate and calcium phosphates. This resorbable material hardens in situ without exceeding body temperature, leading to rapid localized increases in bone mineral density (BMD) (Howe et al. 2020). The injection of AGN1 into osteoporotic hips of human cadavers increased hip strength by an average of 20.5% within 24 hours (Stroncek et al. 2019). After hardening, AGN1 is gradually replaced by new bone matrix that is remodeled and integrates with native bone (Howe et al. 2020; Shaul et al. 2021), leading to significant early (+41% at 12 weeks) and long-term (+22% at 5-7 years) increases in hip strength in women with postmenopausal osteoporosis based on finite element analysis (Howe et al. 2020).
LOEP is included among treatment options for patients with osteoporosis at very high fracture risk in the most recent European guidance for the diagnosis and management of osteoporosis in postmenopausal women (Kanis et al. 2018), and AGN1 LOEP can also be performed as an adjunct to systemic osteoporosis pharmacotherapy (Howe et al. 2020). The ability of LOEP to reduce secondary hip fractures in postmenopausal women with osteoporosis is currently under investigation in the large Phase 3 RESTORE trial (ClinicalTrials.gov: NCT04796350). The commercial availability of AGN1 for LOEP in several European countries provided an early opportunity to evaluate real-world surgeon experience and patient satisfaction with the procedure, which are important determinants of product acceptance beyond the forthcoming efficacy results from the RESTORE trial.
The current study, conducted at a community-based orthopaedic surgery practice in Belgium, provides detailed, real-world results on the administration, experience, satisfaction, performance, regional bone density changes, and safety of AGN1 LOEP in one or both hips of 43 women and men at high risk of hip fracture based on low hip BMD T-scores and/or clinical risk factors including prior fragility fractures. The study hypothesized that the LOEP treatment would be well-tolerated and have high levels of participant satisfaction.
Materials and Methods
The GRACE study is an open-label, retrospective, non-randomized, non-blinded, single-surgeon, single-center study involving participants previously treated with AGN1 LOEP (Clinicaltrials.gov: NCT05966974). Participants were eligible to participate if they received AGN1 LOEP treatment between September 2019 and November 2022, were willing to participate in the study, and able to give written informed consent. Participants were ineligible if their AGN1 LOEP treatment was previously performed as part of a protocoled clinical study. The clinical investigation plan was approved as EC22057 by the Ethics Committee of the Vitaz Hospital, and conducted in accordance with the United States Code of Federal Regulations ISO14155, ICH Good Clinical Practices, and the Declaration of Helsinki (2013).
A single post-LOEP follow-up evaluation was conducted at the time of enrollment, comprising a medical record review and completion of a prospective patient questionnaire. The full list of assessments is in Supplemental Table S1. The primary study objective was to evaluate safety, participant satisfaction, and performance, the latter based on incidence of fragility fractures of treated hips. The population for analysis comprised participants treated with AGN1 LOEP, and per-protocol and safety populations are identical because no deviations occurred within the study.
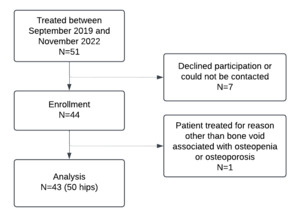
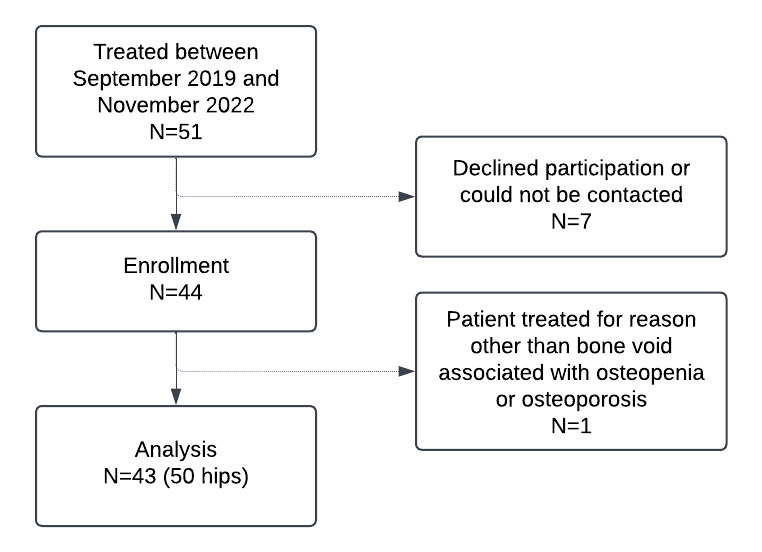
Figure 1 provides a visual depiction of eligibility and enrollment. Fifty-one participants were treated with AGN1 LOEP between September 2019 and November 2022, 44 of whom enrolled in the study and had a total of 52 hips treated. Five patients chose not to enroll and two patients could not be contacted to obtain consent. One participant was treated for reasons other than bone voids associated with osteopenia or osteoporosis and was excluded from this analysis, leaving a final N of 43. These 43 participants had 50 hips treated for bone voids associated with osteopenia or osteoporosis and were included in the analysis. Table 1 describes demographics and baseline characteristics. Average age was 64.5 ± 13.1 years, and 93.0% of participants were female. Based on FRAX[1], the mean baseline 10-year risk of MOF was 20.5 ± 9.5% and the mean 10-year risk of hip fracture was 7.5 ± 5.5%. Hip fracture risk by FRAX exceeded 3% for 37 participants and exceeded 4.5% for 28 participants. Most treated participants (37/43) had a history of fragility fracture, and the average number of prior fractures in the overall population was 2.7 ± 3.0. 32.6% (14/43) had a hip fracture history and 23.3% (10/43) participants had a spine fracture history. Baseline DXA, available for 47 of 50 treated hips (Supplemental Table S2), indicated mean BMD values of 0.707 ± 0.055 g/cm2 for the femoral neck corresponding to mean BMD T-Scores of -2.39 ± 0.39.
Baseline (pre-LOEP) DXA of the treated hip revealed that 19/43 participants (44.2%) had osteoporotic femoral neck BMD T-scores and 23/43 (53.5%) had osteopenic femoral neck T-scores. One participant treated bilaterally had one femoral neck with a baseline T-Score of -2.1 (osteopenic) and the other -2.6 (osteoporotic). The subgroup with osteoporotic T-scores had a mean age of 66.1 ± 13.5 years and a mean hip FRAX score of 10.9 ± 6.5, and the subgroup with osteopenic T-scores had a mean age of 63.2 ± 13.1 years and a mean hip FRAX score of 4.8 ± 2.2. Forty of the 43 participants met one or more criteria for very high fracture risk based on contemporary clinical practice guidelines (Camacho et al. 2020; Cosman et al. 2014; Shoback et al. 2020). These criteria include hip FRAX score >4.5 (28 participants) (Camacho et al. 2020), MOF FRAX score >30 (4 participants) (Camacho et al. 2020), multiple prior fractures (23 participants), multiple prior spine fractures (2 participants) (Camacho et al. 2020; Cosman et al. 2014; Shoback et al. 2020), very low BMD T-score (< -3.0) (3 participants) (Camacho et al. 2020), osteoporotic BMD T-score with a fracture history (22 participants) (Shoback et al. 2020), and osteoporotic BMD T-score with a hip fracture history (8 participants) (Cosman et al. 2014). The other 3 participants met criteria for high fracture risk based on participant age and hip FRAX scores >3% (Camacho et al. 2020). A list of high or very high fracture risk criteria met by each participant is provided in Supplemental Table S3.
All participants (43/43) were on either a bisphosphonate (37/43), denosumab (9/43), teriparatide (1/43), or romosozumab (1/43) at some point in their medical histories. Thirty-two of the 43 participants were on osteoporosis pharmacotherapy at the time of LOEP; the timing of osteoporosis therapy in relation to LOEP was unknown for 2/43 participants (4.6%). At the time of medical review, 40/43 participants were undergoing osteoporosis therapy. Participants had ongoing prescriptions for bisphosphonates (30/43), denosumab (9/43), or romosozumab (1/43).
The AGN1 LOEP Kit is a CE Marked product in the European Economic Area, Switzerland, and UK. The AGN1 LOEP Kit is not currently marketed in the United States or anywhere else outside of Europe.
AGN1 LOEP treatment was performed on supine patients on a fracture table with C-arm fluoroscopy. The procedure involves injection of implant material, a triphasic, resorbable implant material consisting of calcium sulfate, brushite, and β-tricalcium phosphate, into a surgically prepared site in the proximal femur. The site was accessed by making a small incision in the lateral thigh followed by introduction of a guide pin and a 5.3 mm cannulated drill through the lateral femoral cortex to the base of the femoral head. The centrally located enhancement site was gently debrided followed by aspiration and saline irrigation to remove fat and other non-structural elements. The implant material was then mixed and injected under fluoroscopic guidance to backfill the enhancement site from the apex to the cortical access portal.
Technical success of AGN1 LOEP was assessed by a questionnaire provided to the principal investigator (PI) regarding the ability to deliver the necessary amount of implant material to the enhancement site. Participant satisfaction was collected using a visual analog scale (VAS) Satisfaction score (range, 0-100 mm). Safety evaluation consisted of reviewing the incidence of all adverse events (AEs) and serious adverse events (SAEs) determined to be at least possibly related to the procedure and/or device post-treatment.
All additional evaluations were completed through a medical record review focused on demographic information, medical history, medications, and supplements related to osteoporosis, pain, and use of glucocorticoids. Surgical record review included implant material volume, incidence of fragility fractures, timed up and go (TUG) measurements, Parker Mobility scores, Forgotten Joint scores, review of available radiographic data, rehabilitation records, and review of any additional quality of life measures collected as part of routine care. Data were collected and analyzed for each of the previous visits related to the prior AGN1 LOEP treatment.
The estimated 10-year risk of hip fracture and major osteoporotic fractures (MOF, i.e., clinical spine, forearm, hip, or shoulder fractures) was assessed pre-operatively with the online fracture risk calculator FRAX® (Kanis et al. 2009). Baseline and follow-up BMD values by dual X-ray absorptiometry (DXA) were collected for descriptive statistics. Medical records were reviewed for any fragility fractures before and after treatment.
TUG scores reflect the time required to rise from a chair, walk three meters, turn around and walk back to the chair and sit down (Laflamme et al. 2012). If multiple TUG measures were available, descriptive statistics of the change from baseline were presented for the last visit. Quality of life (QOL) measures comprised Parker Mobility Scores and Forgotten Joint Scores. Parker Mobility Scores were based on 3 questions worth up to 3 points each, for a range of 0 (worst mobility) to 9 (best mobility), regarding participants’ ability to move around the home, get out of the home, and go shopping (Voeten et al. 2020). The Forgotten Joint Score instrument involves 12 equally weighted questions regarding participants’ conscious awareness of a target joint during activities of daily living. Responses, each on a 5-point scale, are transformed for a total range of 0 to 100 points, with higher scores reflecting better outcomes (Behrend et al. 2012).
Pain was assessed using the 100 mm VAS. Participants underwent VAS pain assessments at each evaluation to indicate their hip/thigh pain for each treated hip. If multiple measures were available, descriptive statistics of the change from baseline were presented for the last visit. All incidents of prescriptive rehabilitation were collected via review of medical records for all participants.
All post-operative radiographs were assessed by the PI for resorption of the implant material and its replacement with bone. Resorption was assessed in 25% increments, with 0% resorption defined as 100% residual implant material on the initial post-operative X-ray, and complete (100%) resorption defined as 0% residual implant material remaining in the initially filled enhancement site. The PI also recorded whether or not resorbed implant material was successfully replaced with new bone, with new bone defined as having radiodensity at least as great as the surrounding trabecular bone with evidence of bone remodeling.
Descriptive statistics were used to evaluate data collected during the study such as participant characteristics and participant disposition. As the study was not intended to test any a priori stated statistical hypotheses of AGN1 LOEP performance or safety, no statistical inference was performed. Data calculations were performed in Microsoft Excel. All listed variance values reflect standard deviations unless otherwise noted.
Results
All 43 participants were treated with AGN1 LOEP under general anesthesia, comprising 36 participants (83.7%) who underwent unilateral LOEP, 4 participants (9.3%) who had both hips treated with LOEP in separate surgical sessions, and 3 participants (7.0%) who had both hips treated in a single operative session (Table 2). Participants who had one LOEP treatment in a single procedure had a mean anesthesia time of 39.8 ± 17.0 minutes, with an average skin-to-skin time of 14.3 ± 1.9 minutes. Participants who had both hips treated in a single procedure had an average anesthesia time of 64.7 ± 15.5 minutes, with a skin-to-skin time of 11.5 ± 0.7 minutes per hip. Additional procedural characteristics are shown in Table 2. Technical success of the procedure was confirmed in all 50 (100%) of treated hips based on successful delivery of the necessary volume of implant material. The volume of implant material delivered to the enhancement site was 14.8 ± 3.8 mL for osteopenic hips and 16.2 ± 3.3 mL for osteoporotic hips.
Of the 50 hips treated with AGN1 LOEP, 10 participants and 11 hips had both pre- and post-operative DXA scans available for review. Post-operative DXA performed on average 397 ± 216 days after LOEP indicated mean increases from baseline in femoral neck and total hip BMD of 28% ± 19% and 25% ± 15%, respectively (Supplemental Table S4).
The results of patient-reported outcome (PRO) measures are provided in Table 3. Pre- and post-operative TUG test results were available for 39 participants. The mean pre- and post-operative TUG times were 10.6 ± 7.4 seconds and 9.1 ± 3.5 seconds, respectively, reflecting a mean improvement of -1.6 ± 6.6 seconds. Pre- and post-operative Parker Mobility scores were available for 43 participants. The mean pre- and post-operative mobility scores were 8.3 ± 1.6 and 8.4 ± 1.6, respectively, reflecting a mean absolute change of 0.1 ± 0.6.
Pre- and post-operative Hip Forgotten Joint Scores were available for 35 participants. Mean pre- and post-operative joint scores were 46.4 ± 37.1 and 74.5 ± 32.1, respectively, reflecting a mean absolute improvement of 28.1 ± 37.7. Of the 35 participants with available data, 22 (62.9%) improved their joint score, 7 (20.0%) had no change, and 6 (17.1%) decreased their score. Pre- and post-operative VAS Pain Scores were available for 40 participants with a total of 46 treated hips. The mean pre- and post-operative pain scores (mm) were 32.6 ± 30.3 and 13.4 ± 22.8, respectively, reflecting an absolute improvement from baseline of -19.2 ± 32.8.
Of the 47 surgeries performed on 43 participants and 50 hips, average time to hospital discharge from procedure was 0.8 ± 0.4 days. Following LOEP, 12 of 43 participants (27.9%) underwent postoperative physical rehabilitation, primarily for pre-existing conditions, deconditioning unrelated to LOEP, and pain or gait issues.
Participants generally reported a high degree of satisfaction with AGN1 LOEP, with a mean satisfaction score of 81.3 ± 25.3 (maximum = 100) and a median score of 87.0 (interquartile range, 80.0, 97.0). The results of a detailed prospective participant questionnaire are provided in Table 4, with each response rated on a scale of 0 (least favorable impressions) to 5 (most favorable). Mean scores for this 13-item questionnaire ranged from 3.7 ± 1.0 (“I was satisfied with my osteoporosis treatment prior to LOEP treatment”) to 4.6 ± 1.0 (“Would you recommend this surgery to someone else?”).
Table 5 provides a summary of AEs within the study. A total of 21 AEs were recorded among the 43 participants, of which 17 (81%) AEs recovered or resolved and 4 (19%) were ongoing at the end of the study. Of the ongoing adverse events, three were not related to the device or procedure (pain in the knee following total knee arthroplasty, shoulder fracture, persistent spinal pain syndrome) and one had causal relationship to the procedure (pain and discomfort at site related to post-operative seroma). Most AEs were mild (13/21, 61.9%) and 8/21 AEs (38.1%) were serious AEs (SAEs). No AEs were fatal. Most AEs (17/21, 81%) were considered not related to the study device, and (13/21 AEs (61.9%) were considered not related to the study procedure (LOEP). No AEs (0%) were caused by device malfunction or damage. The primary safety endpoint was the percentage of participants with AEs that were at least possibly related to the device or procedure, which was 7/43 (16.3%; 95% confidence interval 6.8%, 30.7%) with one participant experiencing two possibly related AEs.
A list of related AEs is found in Supplemental Table S5, all of which were considered causally related to the procedure and, in some cases, at least possibly related to the device. Seven of the 8 possibly related AEs recovered or resolved during the study, whereas one such AE (pain and discomfort at incision site) was ongoing at study exit. In this case, the participant experienced a post-operative seroma that may have led to pain and discomfort.
Two participants (2/43) experienced a fragility fracture (one a hip, the other a shoulder) following AGN1 LOEP treatment. A 75-year-old woman experienced a fracture of the LOEP treated hip following a fall while getting dressed. This participant had 3 previous fractures (hip, wrist, and pelvis), femoral neck T-scores of –3.2, and FRAX hip fracture risk of 31.0%. The hip fracture occurred 64 days after LOEP and was adjudicated by the PI as not related to the device or procedure; a total hip replacement was performed without complication. Based on the number of years since LOEP for all patients, there was one hip fragility facture in 62.6 patient-years. The second post-operative fragility fracture was in a participant who experienced a shoulder fracture following a fall. This participant had a history of 3 fragility fractures (hip, vertebrae, and pelvis) prior to AGN1 LOEP treatment.
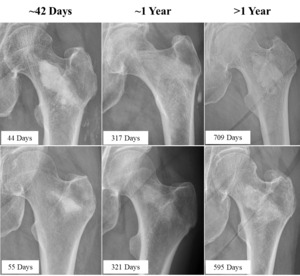
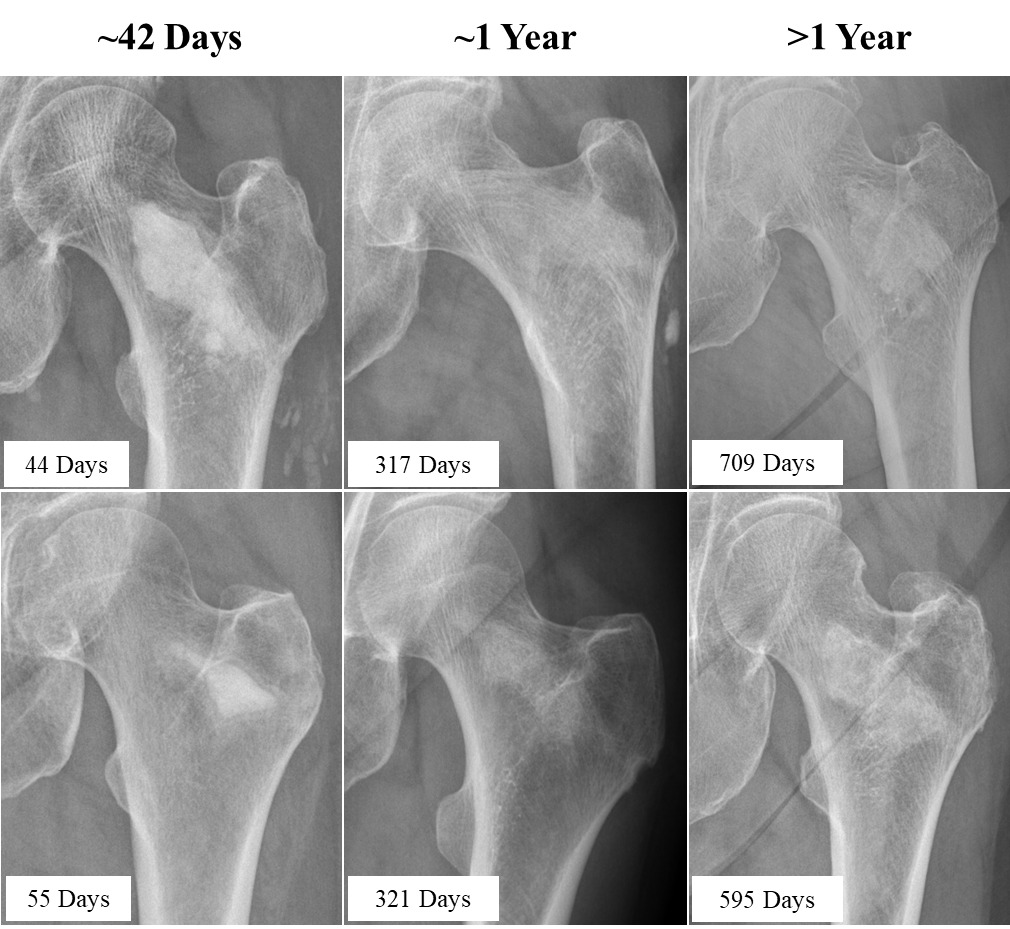
The PI evaluated implant resorption and regional new bone formation on post-LOEP radiographs that were available for 26/50 treated hips; these radiographs were obtained an average of 311 ± 280 days after LOEP (range, 23 – 842 days). Radiographs from six representative patients at three different post-operative time points are highlighted in Figure 2. The mean area of high radiodensity associated with the residual material was 29% ± 40% compared to the post-injection fluoroscopic images, with a median value of 0% (interquartile range 0%, 75%) (Supplemental Table S6). Twenty-five of 26 hips (96.2%) showed radiodensity consistent with new local bone formation, and the single hip film that did not was obtained just 23 days post-LOEP.
Discussion
This study shows that AGN1 LOEP was successfully performed on osteopenic and osteoporotic hips of patients at high risk of hip fracture, with the surgeon able to deliver the necessary amount of implant material to fill the site in osteoporotic and osteopenic hips alike. Participants tolerated the treatment and mobilized within a day on average. Nearly all participants were treated with systemic osteoporosis therapies, which did not require alteration due to LOEP treatment. Participants expressed satisfaction with their treatment outcomes, with most reporting they would undergo LOEP again and would recommend it to others. A high proportion of patients treated with LOEP were satisfied that the treating physician discussed the treatment with them and that such discussions supported their favorable opinion of the treating physician. The safety profile was generally consistent with that of other minimally invasive orthopedic hip procedures performed in an elderly population (Trojani et al. 2012; Hitos and Fletcher 2009; Pedersen et al. 2014; Kaneko, Hirakawa, and Fushimi 2014; Anakwe et al. 2012; Blomfeldt et al. 2006).
The average baseline hip FRAX score for this population was 7.5%, well above the 4.5% score that identifies patients at very high risk of fragility fracture (Camacho et al. 2020). That observation, combined with a history of osteoporosis pharmacotherapy use by all 43 participants and a fragility fracture history for 37 participants, indicates that the use of AGN1 LOEP in this study was directed toward and accepted by patients with appropriately high hip fracture risk. Evidence that 93% of participants were on systemic osteoporosis therapy at the time of medical review (415 ± 337 days post-LOEP) suggests that AGN1 LOEP does not reduce initiation or compliance with other osteoporosis treatments.
Marked post-LOEP increases in femoral neck and total hip BMD were observed among the substantial subset of participants with pre- and post-LOEP DXAs, with the latter scans obtained an average of 397 days post-LOEP. Radiographic evaluation indicated substantial implant resorption after a mean follow-up of 311 days, at which time 96% of all treated hips exhibited radiodensity at the LOEP site consistent with that of normal trabecular bone. These findings corroborate previous evidence of new bone formation and substantial long-term (5-7-year) BMD gains in AGN1 LOEP-treated hips of women with postmenopausal osteoporosis (Howe et al. 2020). VAS Pain Assessment scores and Forgotten Joint Scores improved from pre-LOEP to post-LOEP assessments, with both PRO measures improving to degrees that are considered clinically significant (Bird and Dickson 2001; Robinson et al. 2021). These PRO findings should be interpreted with caution due to the open-label nature of the study.
The ability of AGN1 LOEP to reduce hip fracture risk has yet to be evaluated in suitably powered trials, but the procedure increases biomechanical properties of osteoporotic hips in cadavers within 24 hours (Stroncek et al. 2019). Further, a randomized controlled trial is currently ongoing to address this question (NCT: NCT04796350). Approximately one third of the current participants had a history of hip fracture, and numerous clinical practice guidelines and expert opinions advise that a hip fracture history is sufficient to diagnose very high fracture risk without confirmatory DXA (Camacho et al. 2020; Cosman et al. 2014; Shoback et al. 2020; “Management of Osteoporosis in Postmenopausal Women: The 2021 Position Statement of The North American Menopause Society” 2021; Kanis et al. 2021). Such guidance supports the potential for contralateral LOEP treatment during the same operative session as hip fracture surgery, which may represent an efficient use of surgery resources that assures LOEP fulfillment for patients at high risk of another hip fracture (Adachi et al. 2021; Hannan et al. 2019; Johansson et al. 2017). Recent analyses of >115,000 patients with an incident hip fracture shows that the most common second fracture was another hip fracture, which comprised 33% of all second fractures after a median interval of just 1.5 years (Schemitsch et al. 2022). Other studies show that an index hip fracture carries a 3-4-fold increase in the relative risk (Gehlbach et al. 2012; Hagino et al. 2012) and a 9-12% in the absolute risk of another hip within 1-2 years (Ryg et al. 2009). Unfortunately, current pharmacotherapies have not been proven to reduce the risk of second hip fractures, even in a large trial wherein a potent pharmacotherapy was initiated within 90 days of surgical hip fracture repair (Lyles et al. 2007). This evidence gap may partly contribute to the low and potentially declining rates of osteoporosis pharmacotherapy among patients with recent hip fracture (Tajeu et al. 2014; Hagino et al. 2012; Galli et al. 2022; Kamel et al. 2000; Kim et al. 2016; Rabenda et al. 2008; Taylor et al. 2021).
Among this study’s strengths is that it reflects real-world use of AGN1 LOEP in a community-based orthopedic practice. Another strength is that all participants had high or very high fracture risk at the time of LOEP based on BMD T-scores, fracture history, and FRAX scores (Trost et al. 2020). Study limitations include its retrospective, open-label, single-surgeon, single-center design, as well as its reliance on non-standardized intervals from LOEP to enrollment, potentially introducing recall bias. Another limitation is that post-operative rehabilitation reasons were collected, but their relatedness to the procedure was not documented and may have been related, in some cases, to co-morbidities among the study participants, some of whom reported pre-existing lumbar pain, stroke, and/or muscle atrophy.
This study supports a favorable benefit-risk for AGN1 LOEP treatment in a real-world study population at risk of hip fragility fracture when used to treat bone loss in the proximal femur associated with osteopenia or osteoporosis. Based on the results of this study, the LOEP treatment is worthy of consideration for treatment of bone voids in the proximal femur associated with osteopenia or osteoporosis.
List of Abbreviations:
-
AGN1: Triphasic calcium-based, resorbable implant material
-
AE: Adverse event
-
BMD: Bone mineral density
-
DXA: Dual X-ray absorptiometry
-
ICH: International Council for Harmonisation of Technical Requirements of Pharmaceuticals for Human Use
-
ISO: International standards organization
-
LOEP: Local osteo-enhancement procedure
-
MOF: Majority osteoporotic fracture
-
PI: Principal investigator
-
QOL: Quality of life
-
SAE: Serious adverse event
-
TUG: Timed up and go
-
VAS: Visual analog score
Acknowledgments
Editorial assistance was provided by Paul Kostenuik, PhD (Phylon Pharma Services).
Funding
This study was funded by AgNovos Bioscience.
FRAX is a widely used fracture risk assessment tool (https://frax.shef.ac.uk/FRAX/)