Introduction
Orthopedic surgeries such as total knee arthroplasty (TKA), rotator cuff repair, and anterior cruciate ligament (ACL) reconstruction provoke significant physiological stress marked by elevated cytokines (IL-6, TNF-α) and acute-phase reactants such as C-reactive protein (CRP) that correlate with pain, muscle atrophy, and delayed mobilization. Excessive or prolonged inflammation has been linked to slower rehabilitation, impaired wound healing, and increased risk of complications (Hall et al. 2001; Rossaint and Zarbock 2018; Barretto et al. 2017; Huang et al. 2020).
Current pharmacologic strategies, which include NSAIDs, corticosteroids, and opioids, carry limitations including gastrointestinal toxicity, impaired bone healing, and dependency risk (Zhao-Fleming et al. 2018; Buchheit, Holt, Sheu, et al. 2018). This underscores the need for safe, non-pharmacologic adjuncts that can modulate inflammation and oxidative stress without impeding recovery. Tart cherries (Prunus cerasus), particularly Montmorency varieties, are rich in anthocyanins, flavonols, and phenolic acids, compounds known to inhibit cyclooxygenase (COX-1 and COX-2) and nuclear factor-κB (NF-κB) signaling. Thereby, they can help in reducing prostaglandin-mediated inflammation (Seeram et al. 2001; Kelley, Adkins, and Laugero 2018; Blando, Gerardi, and Nicoletti 2004). Several clinical trials in non-surgical populations demonstrate reductions in CRP, IL-6, and oxidative stress markers following tart cherry supplementation (Keane et al. 2016; Kelley, Adkins, and Laugero 2018; Chai et al. 2019).
In addition, tart cherry has shown analgesic and anti-inflammatory effects in patients with knee osteoarthritis, leading to improvements in pain and joint function (Schumacher, Pullman-Mooar, Gupta, et al. 2013; Ducharme, Baribeau, Lalonde, et al. 2016). Comparable findings in exercise-induced muscle damage trials suggest enhanced recovery and reduced soreness (Howatson et al. 2012; Ducharme, Baribeau, Lalonde, et al. 2016; Lima, Oliveira, Santana, et al. 2022; Connolly et al. 2006). Despite these promising data, the role of tart cherry supplementation in postoperative orthopedic recovery remains uninvestigated. Additionally, to our knowledge, no prior review has specifically evaluated tart cherry supplementation within the context of orthopedic surgical recovery. Although surgical tissue trauma and exercise-induced muscle injury share overlapping inflammatory and oxidative pathways, these conditions are physiologically distinct, and mechanistic overlap alone does not establish therapeutic transferability. Tart cherry supplementation should be considered an investigational candidate rather than a therapeutic intervention in postoperative orthopedic recovery. As such, this review aims to evaluate the mechanistic and clinical evidence supporting tart cherry supplementation as a potential anti-inflammatory and antioxidant adjunct to enhance postoperative recovery in orthopedic surgery.
Methods
This manuscript is a narrative review intended to synthesize mechanistic, translational, and indirect clinical evidence related to tart cherry (Prunus cerasus) supplementation and inflammation-mediated recovery pathways. The literature search was exploratory and hypothesis-generating rather than systematic, and no formal study selection hierarchy, quantitative synthesis, or PRISMA-guided methodology was applied. Risk-of-bias considerations were used descriptively to contextualize heterogeneity across studies rather than to rank evidence strength.
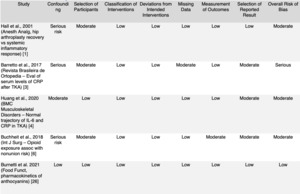
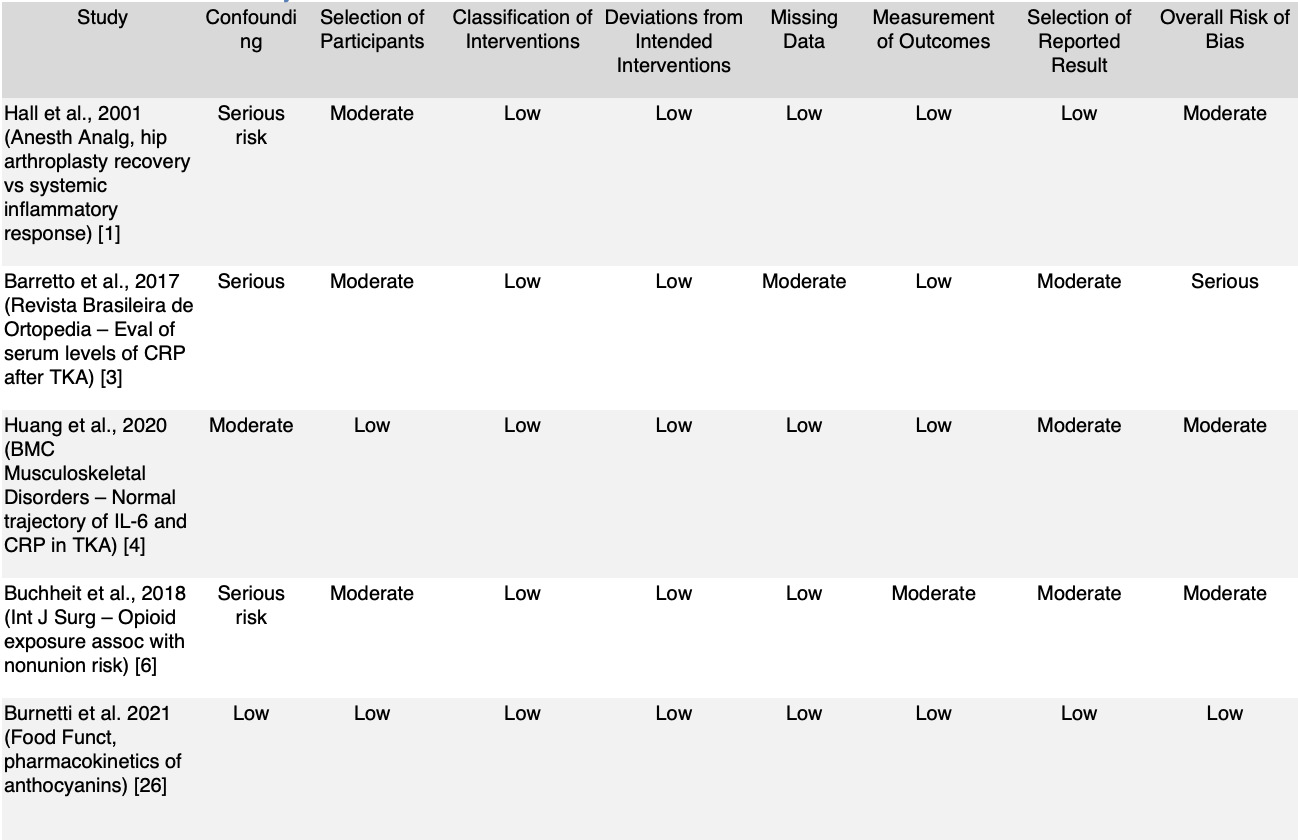
Across included studies, overall risk of bias ranged from low to moderate, with most randomized trials demonstrating adequate randomization, outcome assessment, and retention. Common limitations included small sample sizes, short intervention durations, and heterogeneous dosing protocols. These factors constrain direct clinical extrapolation but do not negate the reproducibility of observed anti-inflammatory and antioxidant effects across populations. Detailed risk-of-bias considerations are provided in Supplementary Tables 1–2.
Mechanistic Rationale
Anti-Inflammatory Signaling Modulation
Tart cherries’ polyphenolic compounds, especially anthocyanins and flavonoids, inhibit the cyclooxygenase (COX-1 and COX-2) enzymes and suppress the activation of nuclear factor kappa B (NF-κB), reducing transcription of pro-inflammatory mediators such as TNF-α, IL-1β, and IL-6 (Seeram et al. 2001; Kelley, Adkins, and Laugero 2018). While these mechanisms overlap with pathways targeted by NSAIDs, such biochemical similarity does not imply comparable clinical efficacy or safety, particularly in perioperative settings (Kelley, Adkins, and Laugero 2018). Clinical studies demonstrate that tart cherry juice significantly reduces circulating C-reactive protein (CRP) and IL-6 levels after acute physiological stress (Keane et al. 2016; Chai et al. 2019). Even though there is inconsistent reporting on vascular benefits, Kimble et al. (2021) showed that 4 weeks of Montmorency tart cherry concentrate did not alter blood pressure or artery stiffness in healthy adults, but profiling of the urine showed biochemical changes of metabolites. This suggests systemic antioxidant and metabolic effects that may precede grossly measurable vascular improvements (Kimble et al. 2021). These biochemical changes have been associated with improved muscle recovery and reduced systemic inflammation in both young and older adults.
Antioxidant and Mitochondrial Protection
Orthopedic surgical trauma triggers ischemia-reperfusion injury and mitochondrial dysfunction, leading to the accumulation of reactive oxygen species (ROS). Tart cherry polyphenols enhance endogenous antioxidant enzyme activity, superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase, thereby reducing oxidative stress and lipid peroxidation (Keane et al. 2016). Anthocyanins have demonstrated high oxygen radical absorbance capacity (ORAC) values and direct scavenging of peroxynitrite radicals, contributing to preservation of muscle integrity post-injury (Traustadóttir, Davies, Su, et al. 2009). These biochemical effects have been directly demonstrated in human studies, including pharmacokinetic analyses by other studies (Brunetti et al. 2023). In this study, standardized tart cherry juice was administered at two doses to adults with gout, demonstrating quantifiable and dose-dependent plasma anthocyanin levels.
These pharmacokinetic findings were linked to downstream activation of antioxidant defenses and concurrent inhibition of inflammatory mediators, including TNF and inducible nitric oxide synthase (iNOS). The examined effects showed that the bioactive metabolites found in tart cherry juice were active in an inflamed patient population and not just in healthy populations. A recent randomized controlled trial by Gholami et al. further suggested that anthocyanin-rich tart cherry supplementation works as a modulator of anti-inflammatory effects instead of as a broad suppressor of cytokines. The study, most pronounced in short-term trials among healthy adults, focused on non-exercise interventions and showed a significant reduction in C-reactive protein but unchanged levels of IL-6 and TNF-α (Gholami et al. 2022). In addition, phenolic acids like chlorogenic acid and quercetin derivatives improve endothelial nitric oxide bioavailability, potentially enhancing local perfusion and wound healing (Ducharme, Baribeau, Lalonde, et al. 2016; Kelley, Adkins, and Laugero 2018). Beyond inflammation, tart cherry compounds may exert central and peripheral analgesic effects.
Pain Modulation and Nociceptive Pathways
Experimental data suggest that anthocyanins modulate transient receptor potential (TRP) channels involved in nociception and inhibit prostaglandin E2 synthesis (Seeram et al. 2001; Kelley, Adkins, and Laugero 2018). In human studies, regular consumption of tart cherry juice has been linked with reduced delayed-onset muscle soreness (DOMS) and lower perceived pain scores following strenuous physical activity (Howatson et al. 2012; Lima, Oliveira, Santana, et al. 2022). These analgesic effects may translate to reduced postoperative pain and opioid consumption in orthopedic patients.
Sleep-Inflammation-Recovery Axis
Sleep also plays a crucial role in musculoskeletal healing. Tart cherries contain endogenous melatonin (approximately 13 ng/g of fruit), which can improve sleep onset, duration, and efficiency (Howatson et al. 2012). Improved sleep quality may enhance tissue regeneration, immune function, and overall recovery, especially after surgery (Kelley, Adkins, and Laugero 2018). In a randomized crossover trial, tart cherry juice increased melatonin levels and improved actigraphy-measured sleep duration and efficiency in adults with insomnia (Pigeon et al. 2010). Notably, recent data discovered Montmorency tart cherry supplementation has been shown to improve objective and subjective sleep parameters in healthy adults (Howatson et al. 2012; Tucker, Kim, Gurzell, et al. 2024). However, similar benefits were not observed in individuals with overweight or obesity at commonly used doses. This suggests that baseline metabolic and inflammatory status may influence responsiveness to supplementation (Tucker, Kim, Gurzell, et al. 2024).This pathway provides an additional indirect mechanism for enhancing postoperative outcomes. Additionally, polyphenols in tart cherry have been shown to enhance vascular function by improving endothelial nitric oxide synthase (eNOS) activity and reducing vascular adhesion molecule expression (Traustadóttir, Davies, Su, et al. 2009; Keane et al. 2016). Improved endothelial reactivity supports oxygen delivery and waste clearance during the recovery process, which are critical for muscle and wound repair after orthopedic surgery. Mechanistic plausibility alone cannot be interpreted as evidence of clinical benefit in surgical populations and should be viewed as hypothesis-generating.
Evidence from Related Populations
Exercise-Induced Muscle Damage and Acute Inflammatory Response
Tart cherry supplementation consistently reduces post-exercise inflammation and muscle soreness. Howatson et al. (2012) found attenuated strength loss and reduced creatine kinase (CK) and IL-6 levels in athletes consuming Montmorency cherry concentrate after resistance exercise. Similarly, Ducharme, Baribeau, Lalonde, et al. (2016) observed faster recovery of isometric strength following intermittent exercise. A meta-analysis by Lima, Oliveira, Santana, et al. (2022) encompassing 14 trials concluded that tart cherry intake significantly lowered muscle pain and biomarkers of damage, confirming its reproducible anti-inflammatory effects.
Osteoarthritis as a Chronic Inflammation Model Relevant to Surgical Recovery
In osteoarthritis (OA), chronic low-grade inflammation contributes to cartilage degradation and pain. Tart cherry juice reduced CRP and improved Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores in two RCTs of knee OA patients (Schumacher, Pullman-Mooar, Gupta, et al. 2013; Ducharme, Baribeau, Lalonde, et al. 2016). Patients reported decreased pain and stiffness alongside improved joint function.
Safety and Tolerability in Active Musculoskeletal Conditions
Additional data have been merging to support the tart cherry supplement’s favorable safety profile and tolerability across many populations. For example, Sinclair et al. (2022) showed that supplementation was well tolerated with stable inflammatory markers and no adverse effects in active adults with patellofemoral pain. Although conducted in populations with relatively low inflammatory burden, these findings indicate that tart cherry supplementation maintains biochemical and vascular stability without adverse immune effects. This could support its feasibility in higher-inflammatory states such as postoperative orthopedic recovery.
Sex-Specific Evidence and Controlled Musculoskeletal Stress in Women
Expanding on these findings, sex-specific evidence that has begun to emerge with targeted work in women, continuing to support perioperative applications. In Ortega et al. (2023), active healthy women exposed to standardized neuromuscular strain while receiving tart cherry extract within the loading and recovery windows of activity. Strength, power, muscle activation, and soreness were followed over a three-day period during which time the supplement was tolerated with no adverse events. Participants demonstrated recovery without destabilizing inflammatory responses, which is important when considering post-surgical application. Jointly, these results demonstrate that tart cherry supplementation can be safely administered in a controlled dosing schedule during defined musculoskeletal stress in women. This supports both physiological compatibility and perioperative feasibility in a historically underrepresented population.
Older Adults and Systemic Inflammatory and Oxidative Stress Modulation
Tart cherry supplementation has been studied in non-athletic and older adult populations for its systemic benefits. In a 12-week trial, Kelley, Adkins, and Laugero (2018) demonstrated significant reductions in CRP, oxidized LDL, and lipid peroxidation products among older adults consuming Montmorency tart cherry juice. Similarly, Chai et al. (2019) found reductions in oxidative DNA damage and improved total antioxidant capacity. These results are directly relevant to older orthopedic patients, who often exhibit elevated baseline inflammation and oxidative stress, predisposing them to slower recovery.
Vascular Function, Endothelial Health, and Recovery Implications
Although less emphasized, tart cherry supplementation has shown improvements in endothelial function and blood pressure regulation (Keane et al. 2016; Traustadóttir, Davies, Su, et al. 2009). By improving vascular tone and reducing endothelial inflammation, these effects may contribute to reduced perioperative vascular complications and improved tissue perfusion during recovery.
Overall Safety Profile and Evidence Limitations
Across all populations studied, tart cherry supplementation has been well tolerated, with no significant adverse effects or medication interactions reported (Kelley, Adkins, and Laugero 2018; Lima, Oliveira, Santana, et al. 2022). Its favorable safety profile further supports its exploration as a perioperative adjunct, particularly in multimodal recovery protocols that seek to minimize pharmacologic toxicity. However, many of these trials were limited by small sample sizes, short intervention durations, heterogeneous dosing protocols, and the potential for placebo effects, which constrain direct clinical extrapolation.
Discussion
Key Summary
This review identifies a clear translational opportunity to evaluate tart cherry supplementation in the context of orthopedic surgery. The postoperative period is characterized by a pronounced inflammatory surge driven by tissue trauma, ischemia, and muscle disuse. Interventions that temper this response may facilitate faster mobilization, decrease pain, and reduce dependence on opioids (Rossaint and Zarbock 2018). Although the physiological stress of surgery differs from athletic exertion, both trigger oxidative and inflammatory cascades that tart cherry may mitigate. These convergent mechanisms position tart cherry supplementation as a modulator of postoperative inflammatory burden rather than a broad immunosuppressive agent (Seeram et al. 2001; Kelley, Adkins, and Laugero 2018; Keane et al. 2016; Kelley, Adkins, and Laugero 2018; Chai et al. 2019; Hariri, Amirkalali, and Gholami 2024). Together, findings from Kimble et al. and Gholami et al. suggest that tart cherry supplementation induces early biochemical and inflammatory shifts that may precede overt clinical or vascular changes, particularly in lower-stress populations (Kimble et al. 2021; Gholami et al. 2022). With work done by Brunetti et al. (2023), it is shown that anthocyanin exposure from tart cherry can be characterized because it is shown to achieve predictable plasma levels while directly driving antioxidant effects and downregulating inflammatory signaling. This is important because tart cherry is not just thought of as theoretically anti-inflammatory, but instead it is a quantifiable biochemical intervention. Even in studies involving milder and more localized pain, supplementation showed metabolic homeostasis and good compliance, providing evidence for its application in long-term recovery settings (Sinclair et al. 2022).
A lack of adverse events and maintenance of baseline metabolic processes supports its future use in postoperative protocols. Likewise, data that is women-specific from Ortega et al. (2023) show that scheduled tart cherry extract dosing is attainable, well-tolerated, and compatible with monitoring during recovery in a controlled setting. Moreover, it is important to note that the improvement of tart cherry supplementation can be made on both objective and subjective sleep quality parameters. These improvements were shown to emerge alongside decreased levels of IL-6 and TNF-α (Tucker, Kim, Gurzell, et al. 2024). Improved sleep quality represents an indirect but clinically relevant recovery pathway, as sleep-mediated reductions in inflammatory signaling may amplify postoperative tissue repair and functional recovery.
Notably, tart cherry supplementation warrants investigation within multimodal pain management frameworks, rather than clinical adoption, given the absence of perioperative efficacy and safety data (Shenouda et al. 2022). Improved sleep from tart cherries’ natural melatonin content (Kelley, Adkins, and Laugero 2018) may further enhance recovery quality.
Why Tart Cherry vs Other Nutraceuticals
Tart cherry provides a bioavailable polyphenolic matrix supported by multiple human randomized controlled trials, unlike more common antioxidant supplementation, such as vitamins C and E, for example (Hollman et al. 2011; Meulmeester et al. 2022; Howatson et al. 2012; Brunetti et al. 2023). Anthocyanins and related phenolics from tart cherry have demonstrated measurable plasma concentrations, reproducible reductions in C-reactive protein, and favorable safety profiles across diverse populations, including athletes, older adults, and patients with osteoarthritis (Howatson et al. 2012; Brunetti et al. 2023; Sinclair et al. 2022). Additionally, tart cherry uniquely contains endogenous melatonin, offering a dual anti-inflammatory and sleep-modulating mechanism not shared by most nutraceuticals (Howatson et al. 2012; Pigeon et al. 2010; Tucker, Kim, Gurzell, et al. 2024). These characteristics distinguish tart cherry from antioxidant strategies that lack clinical translatability and support its prioritization for perioperative investigation.
Gaps in Research
However, several gaps continue to remain. No clinical trials have investigated tart cherry perioperatively; optimal dosing and timing are undefined; and interactions with analgesics or anticoagulants remain theoretical. Moreover, orthopedic patients are often older with comorbidities, differing in metabolism and inflammatory responses compared to young athletes.
Limitations
Access to certain potentially relevant studies was limited due to paywalls and database restrictions, which may have constrained the comprehensiveness of this review. Additionally, several included trials featured small sample sizes and short intervention durations, reducing statistical power and potentially limiting the robustness of their conclusions. The generalizability of findings is further constrained by heterogeneity in study populations, supplementation protocols, dosage forms, and outcome measures. While studies were grouped by mechanistic or clinical subtopics to facilitate synthesis, we prioritized more recent and methodologically rigorous investigations. In some instances, specific themes were only included when at least three relevant studies were available, which, while practical, may have introduced selection bias. Furthermore, most data derive from non-surgical populations such as athletes or patients with osteoarthritis, limiting direct extrapolation to postoperative orthopedic settings. Overall, the limited number of high-quality, controlled trials examining tart cherry supplementation in surgical populations underscores the need for future randomized studies to determine optimal dosing, timing, and clinical impact within perioperative recovery frameworks. The predominance of positive findings in the literature raises the possibility of publication bias, and negative or neutral studies may be underrepresented. Additionally, many included trials were underpowered and conducted over short durations, limiting assessment of longer-term outcomes.
Future Direction
Future studies should focus on elective procedures such as TKA, where inflammation and functional recovery are quantifiable through serial CRP/IL-6 measurements, opioid use, muscle mass, and range of motion. Randomized, placebo-controlled trials using standardized Montmorency tart cherry extract initiated pre-operatively and continued post-operatively, could establish feasibility and efficacy. If successful, tart cherry could represent a safe, inexpensive, and accessible adjunct to enhance recovery, reduce healthcare costs, and minimize pharmacologic side effects.
Conclusion
Tart cherry supplementation demonstrates reproducible anti-inflammatory and antioxidant effects across several non-surgical populations; however, no clinical evidence currently supports its use in postoperative orthopedic recovery. This narrative review highlights mechanistic plausibility and indirect human data that may inform future hypothesis-driven perioperative trials. Until such studies are conducted, tart cherry supplementation should be regarded strictly as an investigational candidate rather than a therapeutic or preventive intervention in orthopedic surgical care.