Introduction
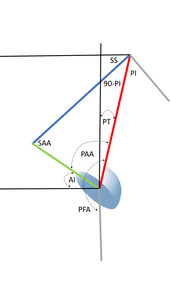
It is important for both hip and spine surgeons to understand the determinants of safe acetabular cup position as procedures in both specialties may substantially alter the orientation and stability of hip replacement components (Lazennec, Brusson, and Rousseau 2011; Esposito et al. 2015; Buckland et al. 2015; Esposito et al. 2018; DelSole et al. 2017; Heckmann et al. 2018; Buckland et al. 2017; Grammatopoulos et al. 2019; Bernstein et al. 2019; Innmann et al. 2022). Research has shown the functional behavior of the hip joint is accomplished through integration between the hip, including the femur and acetabulum, and the lumbar spine as together they coordinate balanced posture in different positions (Heckmann et al. 2018; Ferrero, Liabaud, Challier, Lafage, et al. 2016; Lazennec et al. 2004; Heckmann et al. 2021). The total hip planning paradigm was fashioned upon coronal anatomic reconstruction and until recently did not address the dynamic orientation of the components. Spinopelvic studies have correlated cup orientation with dislocation by measuring sagittal parameters associated with standing pelvic imbalance and mobility abnormalities to a sitting pose (Heckmann et al. 2018, 2021; Grammatopoulos et al. 2022). The biomechanical relationship of sagittal functional cup orientation to pelvic parameter measurements was described by Lazennec (Lazennec et al. 2004), the Anteinclination (AI) cup angle clinically developed by Dorr (Stefl et al. 2017), and recently geometrically modelled (Bodner 2022). The primary relationship employs two horizontally referenced functional angles, the Sacral Slope of S1, (SS), and the sagittal AI cup angle (Kanawade, Dorr, and Wan 2014), to algebraically define an optimizable integrated hip-spine angle, the Sacroacetabular Angle, (SAA) (Lazennec et al. 2004) (Figure 1). However, the hip joint and cup also share an intrinsic functional relationship to the vertically referenced spinal Pelvic Tilt, (PT), defined as the Pelvic Acetabular Angle, (PAA) (Bodner 2022).
Spinal procedures addressing degeneration and deformity may alter both standing PT and functional pelvic mobility when correcting for sagittal imbalance from loss of lordosis in the lumbar spine, thus there needs to be a protocol to analyze if spinal surgery correction will place a hip replacement at risk for instability. This addresses the decision tree as to the proper order of operation in hip-spine patients, which has been based simply on clinical symptomatology or outcomes and not inclusive of understanding the biomechanical effects of one operation on the other. The purpose of this investigation is to expand upon the existing sagittal cup planning methodology employing Sacral Slope sided calculations to define pelvic-cup mechanics through the spinal PT and solely hip-based angles that integrate with spinal surgery planning principles, utilize these parameters in a novel fashion to determine a biomechanically optimum AI cup position, and provide quantitative risk analysis for the potentially deleterious effects of spinal fusion surgeries on established THA cup positions. Secondarily, we attempt to provide a common biomechanical language for clinically relevant planning discussion between the two specialties (Chevarria, Douleh, and York 2021). Finally, we demonstrate the power of a geometrical sagittal hip-spine construct as a biomechanical tool and illustrate 4 applications addressing surgical spinopelvic related questions solvable with mathematical precision.
Methods
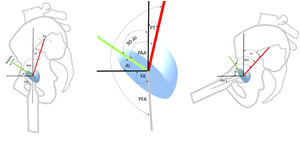
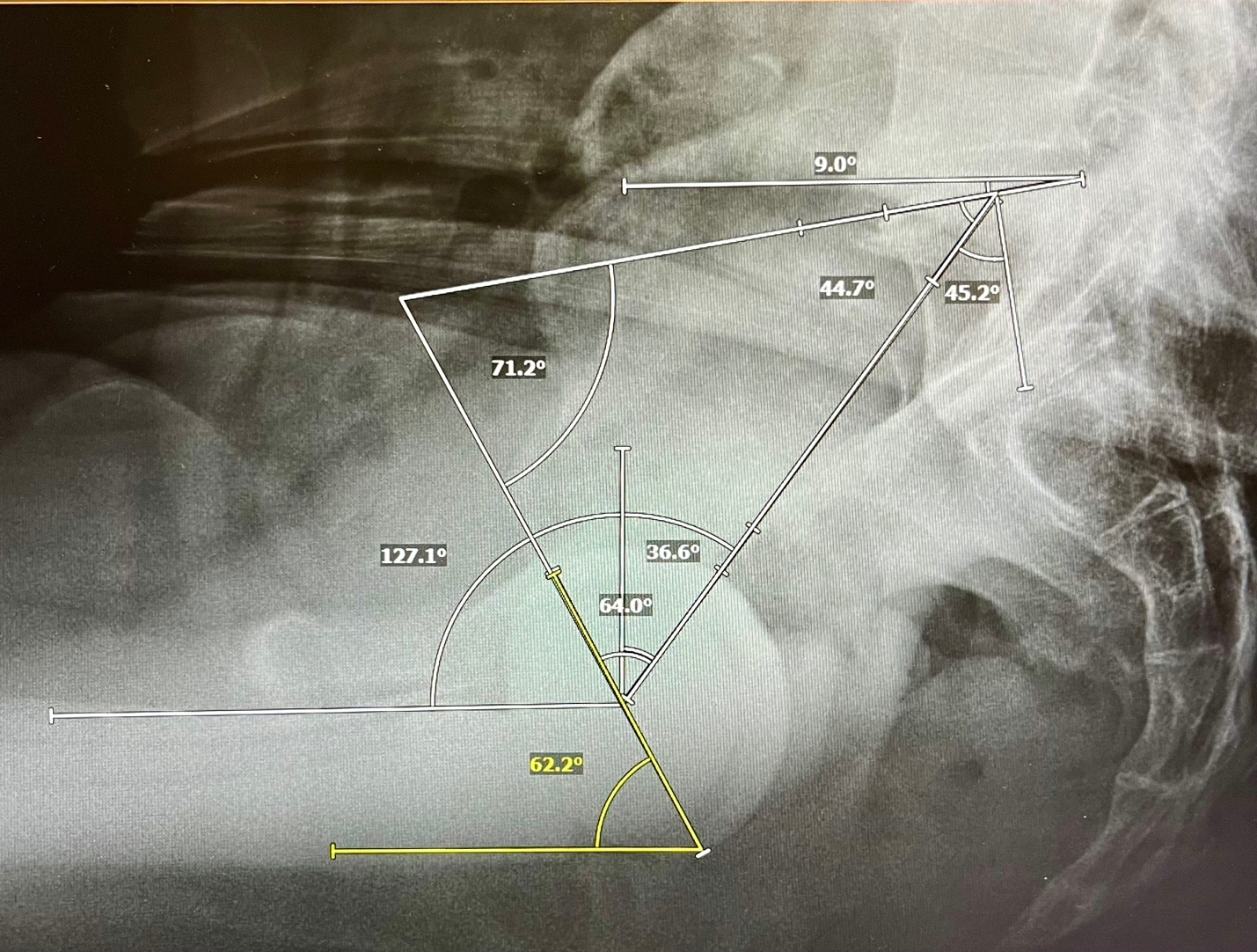
This is a descriptive mathematical study of the sagittal plane functional mechanics of the spinopelvic postural system including a hemispherical acetabular component. The cup may be incorporated into the spinopelvic system using a triangular parameter construct created by combining 2 known angular equations. Firstly, the pelvic parameters are related by the equation defining Pelvic Incidence, PI, a morphologic and functional descriptor of pelvic construction (Legaye et al. 1998). Algebraically, the constant PI equals the sum of the reciprocal functional parameters SS and PT as PI=SS+PT. The acetabular parameters are related by the equation defining the Sacroacetabular Angle of Lazennec, (SAA), as SAA=SS+AI (Lazennec et al. 2004). Superimposing these 2 equations on the sagittal plane of the pelvis reveals the central triangular structure, (Figure 1) connecting three points in space, the anatomic hip and S1 endplate centers and the SAA angle vertex at the intersection of the cup AI tangent line and the SS line (Bodner 2022). The 3 angles created inside this triangle are the anatomically fixed apex angle of (90°-PI), the SAA angle and at the hip joint itself, the Pelvic Acetabular Angle, (PAA), relating the AI cup angle to the PT angle in a similar fashion as the SAA relates the cup to the SS angle. In any individual, SAA and PAA behave reciprocally just as their underlying constituents, SS and PT do. The hip joint combined flexion/extension parameter, Pelvic Femoral Angle, PFA, is measured from S1 center to hip center then continuing parallel to the anterior femoral cortex towards the knee center (Ike et al. 2018).
Applications of the Triangular Model
The integration of the pelvic parameters used in spinal surgery, PI, SS, and PT with the sagittal cup angle AI and the angular relationships of the cup AI to the SS (SAA) and to the PT (PAA) function as a tool to answer different spinopelvic questions. This is accomplished by inserting the angular relationships between the parameters found in any physical situation, such as in the native pelvis where all the mobile parameters are fixed within the pelvic bone, thus SS, PT, AI, even the extraneous APP all move in a 1:1 fashion and PI is a constant. 4 different applications are shown in Figure 3 as the relationships in native anatomy, the normative relationships for any PI value, the relationships for calculating a tilt adjusted AI cup position, and the relationships when spinal surgery repositions the pelvis with a preexisting cup already in place. For simplicity, only the angular ratios for parameters crossing the hip joint associated with the PT side of the pelvis are shown, novel development of PT based parameters is the focus of this work.
Normative values for pelvic and acetabular parameters
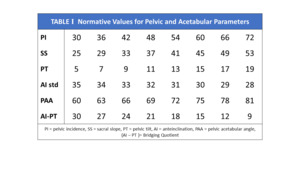
AI, SAA and PAA normative parameters have been adopted using linear equations describing normalized SS and PT relationships for any given PI (Bodner 2022). These equations, SS=0.67PI+5 and PT=0.33PI-5 define that for every 10° change in PI, a normal SS changes 6.67° and PT 3.33°, describing angular ratios of change as 3:2:1 for PI:SS:PT.. Normative values are defined when the cup shares an identical angular relationship to both SS and PT. At this orientation, the triangle’s acetabular angles SAA and PAA become equal at (90+PI)/2 each, this balanced hip-pelvis state creating an isosceles form for the triangle. This value is valid for a static standing position and remains unchanged only when the pelvic mobility between standing and upright seated postures is 25°. The resultant AI cup angle, the sole remaining unknown in the system becomes discoverable as a simple algebraic calculation using either SAA or PAA (Table 1).
The Sagittal Parameters of the Hip Joint
There are 5 angles with their apices at the hip joint that may be analyzed for individualized cup planning or understanding alterations caused by spinal realignment (Figure 2). From superior to inferior, they are PT, (90°-AI), PAA, AI, and PFA. Involuntarily modulating PT represents the body’s main mechanism to balance the trunk and head as they incline forward with aging and deterioration (Barrey et al. 2013; Diebo et al. 2015). PFA represents the collective orientations of the PT angle above and the femoral shaft angle below the hip center, it is intimately correlated to the truncal inclination parameter, the T1 Pelvic Angle, (TPA), thus PFA reactively counterbalances the sagittal spinal imbalance above the hip through flexion/extension of the joint itself (Hasagawa et al. 2016). Adding the calculated AI to the PFA in standing and sitting gives the validated functional safe zone parameter for both acute and chronic dislocation, Combined Sagittal Index, CSI (Heckmann et al. 2018; Grammatopoulos et al. 2022). PAA is algebraically the sum of (90-AI) + PT, the complementary angle to AI is necessary as a consequence of PT being vertically referenced.
Transition from SS based to PT calculation
Functional cup positions have been determined by the spatial position of the pelvis in standing and upright sitting using SS angles measured at the S1 endplate (Kanawade, Dorr, and Wan 2014). Calculations are geometrically simple using the SS as it shares the horizontal reference with AI, (SAA=AI+SS). Determination using the PT requires employing complementary angles including (90-AI), a constituent of the PAA, as again PAA=(90-AI)+PT. Rewriting the equation, PAA=90-(AI-PT) uses the very same bridging cup to pelvis quotient as found in the SAA equation, SAA=PI+(AI-PT).
The quotient (AI-PT) represents a mathematical bridge between the surgically created cup AI angle and the measured PT angle, linking a patient’s intrinsic pelvic morphology and postural compensations to a potentially ideal orientation for the cup. This model has shown a 1° change to PT alters an optimally planned AI target 0.25° to keep it optimal, a ratio of 1:4, an increased PT gives an increased AI (Figure 3). Applying this ratio to the PAA equation solves that for +1° PT change, the PAA angle will increase 0.75°, as PAA reflects the cup’s relationship to the increased PT. SAA decreases reciprocally to preserve the integrity of the central 180° triangle.
Employing PT, cup tilt adjustment is calculated for every 1° the measured PT diverges from the normalized position; in similar fashion to coronal tilt adjustment performed for divergence from a neutral APP pelvic position. The ideal standing AI becomes discoverable by Standing AI= 90-(PAA-PT). Finally, 90-(AI-PT) will confirm the individualized tilt corrected PAA angle and its reciprocal SAA angle.
Functional Mobility Effects on Acetabular Parameters
Functional analysis utilizing the standing and sitting positions requires optimizing the cup’s orientation to an individual’s arc of pelvic excursion as measured by the delta pelvic mobility. Traditionally measured by the Sacral Slope change between standing and sitting, this excursion is identical using PT change. Mobility has a direct effect on the AI, SAA, and PAA angles (Figure 4), the effect increases or decreases the cup’s arc of excursion in similar fashion to opening and closing a hand fan, a 1° change alters each end of the fan by 0.5° with stiffness raising the standing AI and decreasing the sitting AI. Geometrically, mobility of 25° creates an isosceles triangle, the length of the modifiable apex-SS side matching the anatomically fixed S1-hip center side. Stiffness shortens the SS side of the triangle from the equal isosceles length in both postures. Each 1° loss in mobility increases both the SAA and the standing AI angles 0.5° and decreasing the sitting AI 0.5°. Increased mobility has the opposite effect. Mobility effects are constant across all spinopelvic applications, whether calculating for normative values or tilt adjusted cup positions using either SS or PT.
Results
PT based cup calculations- Rule of 4s
Tilt adjusted AI cup positions based on preoperatively measured PI and PT values are demonstrated in Table 2. The ratio of angular change between the cup AI and PT is identical to using SS, 1° to 4°, the correction from normative values may be calculated either using the variance of measured PT from the PI normal values or the PI variance from the PI for which that measured PT would be normal. This Rule of 4s defines when the PT value is 4° greater than the normative PT value for the measured PI, the ideal cup AI orientation will be 1° greater than the normative AI value, and as the PI for any measured PT increases 4°, the ideal cup AI decreases 1°.
Effect of Mobility on Calculated AI cup angles
Table 3 demonstrates the mobility adjusted AI cup angles for normative PT values for any PI. The normative values creating the isosceles triangle condition are those in the 25° mobility row. As explained, a 1° increase in mobility will decrease the standing AI 0.5° for any PI and PT, a 6° increase in PI and 2° increase in PT will decrease the optimal AI 1° in standing. Mobility has the same effect on the AI value in the (AI-PT) quotient; for a given PT, a 1° increase in mobility lowers the AI 0.5° lowering this quotient by 0.5°. This results in increasing the PAA 0.5°, decreasing SAA 0.5° reciprocally.
Solution for tilt adjustment of the (AI-PT) quotient
The relationship between an ideal cup orientation and the changing pelvic tilt creates a quotient of (0.25-1) = -0.75° for each 1° increase in PT (Table 4). A 1° PI increase for any given PT value decreases (AI-PT) by -0.25°. This adjusted quotient may be subtracted from 90° to produce the PAA angle, or added to the PI angle to produce the tilt adjusted SAA angle.
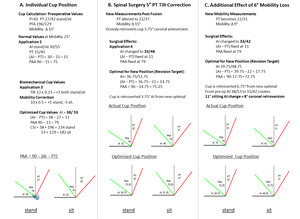
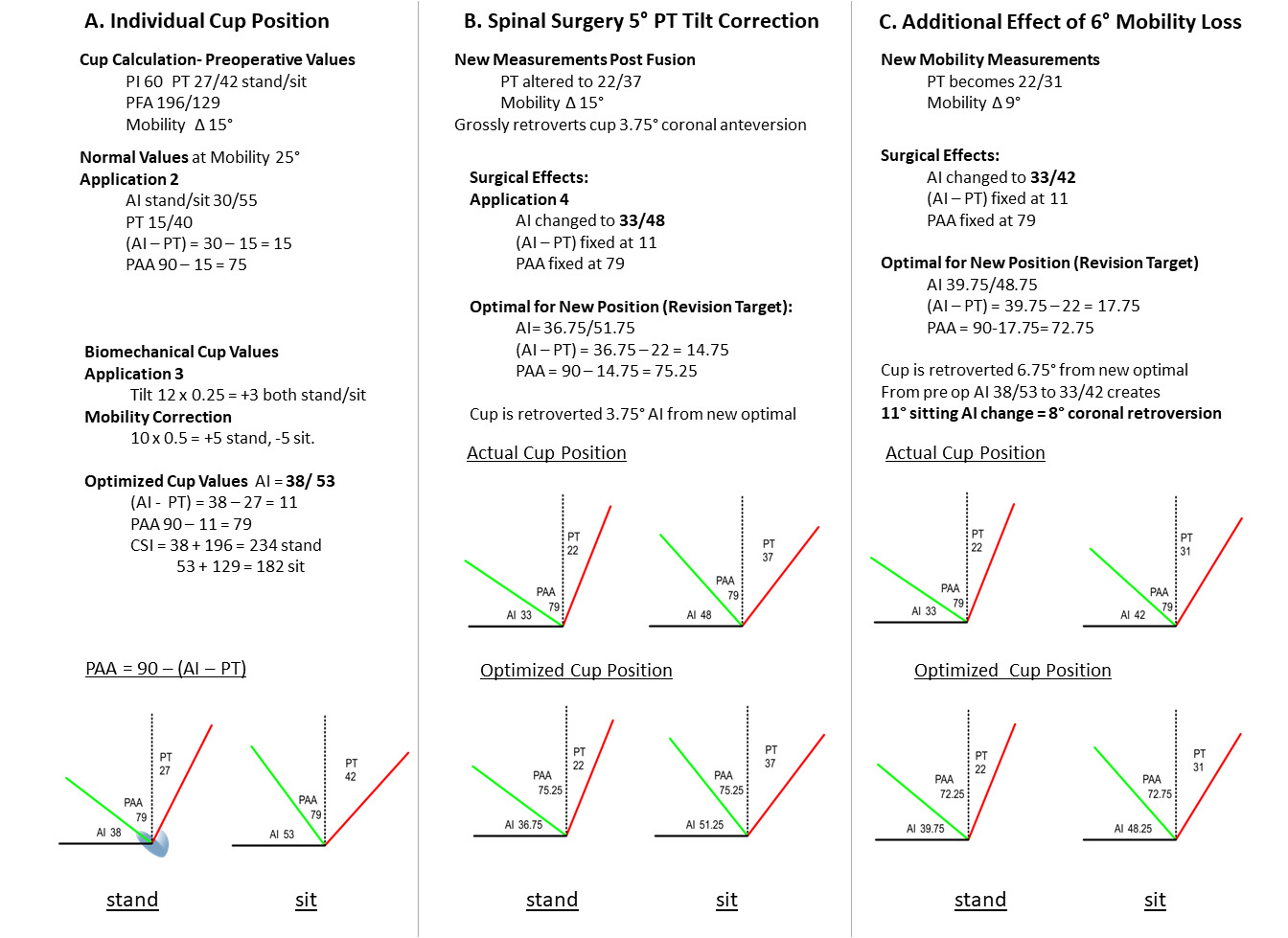
The full sequence of determining biomechanically optimal acetabular parameters, including cup AI, (AI-PT), PAA, and the CSI using PI/PT/PFA preoperative measurements is illustrated in Figure 4, column A.
Effect of Spinal Reconstruction on Cup Orientation
After cup implantation, the SAA and PAA angles become immutably fixed, creating a neo-anatomic state, the fourth spinopelvic construct application (Figure 3(4)). All changes in PT tilt and mobility angles alter AI in a 1:1° fashion instead of the tilt adjustment 4:1° ratio employed in the optimal planning application. For an optimally implanted cup, a 1° decrease in PT then retroverts the cup AI 0.75° (~0.55° radiographic anteversion at inclination of 45°) in both the standing and sitting postures from the postoperative orientation to keep it optimal (Figure 4, column B). Likewise (AI-PT) remains fixed, remaining 0.75° greater than ideal per 1° PT surgical decrease. Additional retroverting effect occurs sitting for each degree of stand/sit pelvic mobility lost from spinal fusions, magnifying this position’s risk. Each 1° loss in mobility retroverts the AI sitting position 0.5° from the ideal orientation, the combined effects of tilt and mobility are additive in sitting while tilt and mobility effects oppose in standing (Figure 4, column C). Thus, a 1° PT correction combined with a 1° loss of mobility retroverts AI 1.25° beyond the new sitting ideal, (~ 0.9° radiographic anteversion loss).
Case Example- Step by step case employing this methodology (Figure 4).
Column A- Calculation of Biomechanically Optimized Cup Orientation
-
Measure patient PI, PT, PFA stand/ sitting: PI 60, PT 27/42, delta SS= 15°, PFA 196/129
-
Compare PT standing to normative PT for PI 60: 27 vs 15, a +12° PT variance
-
Apply 4:1 Tilt correction:
-
+12° PT correction= +3° standing AI correction (Rule of 4s) from normative AI of 30°= 33° AI standing
-
(AI-PT) standing correction = -0.75 x 12= 9° Normative for PI 60= (30-15) =15° 15-9= 6°
-
-
Mobility correction:
- 10° stiffness correction from neutral 25° to measured 15° dSS applies +5° to standing AI, -5° to sitting AI, +5° to (AI-PT). Therefore, the targeted AI becomes 38/53° stand/sit, (AI-PT) becomes 11° making SAA 60+11=71° and PAA 90-11=79°. Sitting (AI-PT) confirms (53-42)=11°.
-
CSI calculation: CSI=AI+PFA stand/sit. Standing limit <244° for posterior impingement, >151° for anterior impingement
Stand- 38+196=234°, Sit- 53+129=182°, this cup is safe within limits.
Column B- Effect of lowering PT 5° through spinal surgery:
- A. PT lowered to 22° without mobility change. The cup AI lowers directly to 33/48° stand/sit. The optimal cup orientation for 22° PT should be 36.75/51.75°, AI delta 3.75° in both postures. The net effective retroversion of the cup is 0.75° lower AI than ideal for every 1° PT is surgically decreased, (0.55° coronal plane anteversion), the cup has been retroverted 3.75° surgically and is now 2° retroverted to its coronal ideal. Once implanted, (AI-PT) remains constant as cup and pelvis now move 1:1.
Column C- Effect of Additional 6° loss of pelvic mobility
- The cup AI becomes 33/42° stand/sit. The optimal orientation should be 39.75/48.75. In toto, cup AI has been altered from initial AI 38/53 ° to 33/42 by the combined spinal surgery effects, 5 and 11° of AI retroversion change, a coronal sitting anteversion loss of approximately 8°, and 5° of coronal retroversion from its new ideal cup orientation.
Discussion
Balanced postures are governed by control of the body’s line of gravity in the sagittal plane, requiring coordinated movements between the spine-pelvis-hip complex (Lazennec, Brusson, and Rousseau 2011; Dubousset 1994; Schwab et al. 2006). Positional changes to sit or bend require considerable mobility between these regions, accomplished through pelvic rotation about the hip joint, spinal shape change, and horizontal translations called shifts (Stefl et al. 2017; Diebo et al. 2016; Ferrero, Liabaud, Challier, Lefage, et al. 2016; Lazennec et al. 2020). As functional awareness grows in both hip and spine surgery, it becomes crucial to develop a common language in which procedures on one side of the pelvis can be understood in terms of effects on the other. This is difficult with the sagittal standing priority in spinal planning and the supine coronal plane paradigm in THA (Masquefa et al. 2015; Liu et al. 2019) APP-derived tilt adjustment in THA planning has been an accepted method of coordinating cup position to that of the pelvis. Intrinsically however, these relationships are quantitatively defined by the equations for the SAA and PAA angles, determining a functionally constant optimal angular relationship of a hemispheric cup to measured or predicted SS and PT values respectively. Postural control has been shown to be modified through PT and PFA, the latter highly correlated with the spinal T1 Pelvic Angle of truncal inclination, (TPA), and PT is an important parameter within corrective spinal planning (Lafage et al. 2009; Protopsaltis et al. 2014). The quotient, (AI-PT) represents a bridge between the tilt and mobility sensitive cup orientation and the functional morphology and orientation of the pelvis, adding (AI-PT) to the PI produces SAA or subtracting it from 90° produces PAA. The key biomechanical concept is the angular ratio of the cup change to pelvic position change differs when planning to place a component freely into the pelvis than when it moves within the pelvis after it is implanted. Acetabular implantation creates new fixed anatomic relationships, functioning as the adjacent segment to the lumbar spine and sensitive to subsequent spinal surgical changes altering pelvic tilt and/or mobility. In a sense, spinal surgery can create an unwitting THA revision of cup orientation and function.
This triangular model allows individualized AI cup calculation through either the SS or PT value, alterations from normative values derived by the tilt adjustment rule of 4s, 4° of SS or PT change will alter an ideally placed cup 1° in both standing and sitting, with mobility’s 2:1 effect of 2° alteration changing standing and sitting AI each 1° in opposing directions. For determining optimal cup position, working through the SS value is simpler geometrically as AI and SS are both measured from parallel horizontal reference lines. But it is the PT value that integrates the hip to the gravity management system, which can be directly used to determine effects on cup position of spinal procedures.
The hip joint is the pivot point for postural pelvic compensatory rotation, the pelvis rotating back adjusting for truncal inclination after which femur and knee flexion can be recruited (Barrey et al. 2011; Lazennec, Brusson, and Rousseau 2013). Anterior pelvic rotation is typically caused by hip flexion contractures. The sum of these compensations may be measured through the PFA hip joint angle as it includes both the PT above and the femoral angle below. A high standing PFA indicates a hyperextended hip, a low sitting indicates a hyperflexed hip, the spinopelvic risk factors for THA impingement leading to instability (Heckmann et al. 2018; Grammatopoulos et al. 2019). The CSI, (AI+PFA), describes how safe optimal cup orientation may be determined through hip-based angles, avoiding impingements from improper cup orientation while including the non-modifiable spinopelvic state (Tezuka et al. 2019). This analysis supports prior work recommending sagittal plane workups in any patient with history of back pain or surgery (Vigdorchik et al. 2019; Grammatopoulos et al. 2023), provides a method of personalized cup orientation based on a patient’s individual biomechanics and provides a basis for development of predictive models for spinal fusion risk as this could provide improved indications for pre-fusion higher constraint acetabular components in at risk primary THA patients.
Discussion regarding sequencing the proper order of operation in patients requiring hip and spine procedures has been framed by the presence of neurological symptoms, flexion contracture, and primary location of pain, there has not been a biomechanical basis to fully interpret the effects or warn of postoperative hip instability following spinal procedures (Malkani et al. 2019; Parilla et al. 2019). Using AI and the SAA/PAA, the effects of lordosis restoring PT reduction on AI cup orientation become quantitative. Spine surgery decreases PT and reduces the mobility of the lumbosacral articulation, both retroverting an implanted cup and increasing the sitting position impingement risk. This PT tilt adjustment retroverts a cup AI 0.75° from ideal for every degree the pelvis is repositioned compounded by any mobility loss leaving the sitting cup AI another 0.5° retroverted for every lost 1° of mobility. However, repositioning an overly anteverted cup in standing may actually improve the construct orientation.
These findings have significant clinical ramification, particularly for spine surgeons planning longer segment lordosis producing spinal fusions (changing spinal tilt) and those crossing the lumbosacral junction (producing pelvic stiffness) in patients with preexisting hip replacements. The sitting position retroversion produced may destabilize a hip that has lower preexisting anteversion, this can be analyzed preoperatively through a sitting lateral view of the pelvis. AI cup angles near the established sitting lower limit of 45° (Tezuka et al. 2019) can stimulate a discussion with the patient or referral to a hip surgeon to discuss risk associated with THA instability. Additionally, if a significant change in PT or multilevel fusion planned, this should be performed prior to a hip replacement and then the hip planned based on the restored pelvic position and residual mobility measures. Integrated planning software automating the risk analysis is the possible future application of this analysis.
The limitations of this analytic model lie in its assumptions and theoretical nature. The adopted equation for the pelvic parameters produces SS/PT values within 1° of published equations derived in spinal literature for the standing position (Bodner 2022). The 25° mobility neutral point defies explanation mathematically but the AI cup positions produced mirror those published by multiple authors (Tezuka et al. 2019; Grammatopoulos et al. 2023). Clinical studies specifically validating this planning algorithm are needed, however due to its sub-1° accuracy, assistive technology would be essential to achieve this type of precision.
In conclusion, spinopelvic relationships linking spine and hip planning parameters may be integrated through geometric representation of the pelvic and acetabular angles. Biomechanically optimal cup orientation may be determined using either the SS or PT angles, and geometry allows understanding the differences between planning for cup position and the effects of changing the orientation of a fixed cup’s position through spinal surgery. Though cup planning is simpler using SS values, it is through the PT side and quotient (AI-PT) that a direct relationship between the functional influences of the two specialties intersect.





_over_the_pi_range._the_quo.png)






_over_the_pi_range._the_quo.png)