Introduction
Adolescent idiopathic scoliosis affects up to 2% of schoolchildren and is increasing in prevalence worldwide (H.-K. Wong et al. 2005; Sung, Chae, Lee, et al. 2021). If left untreated, it can result in chronic pain, deformity, cardiopulmonary compromise, social isolation, and even death (Weiss et al. 2016). Early detection and management, often via bracing or physiotherapy, are crucial and typically rely on serial scoliosis radiographs to monitor progression (H.-K. Wong et al. 2005; Sung, Chae, Lee, et al. 2021; Weiss et al. 2016).
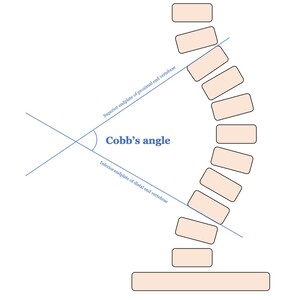
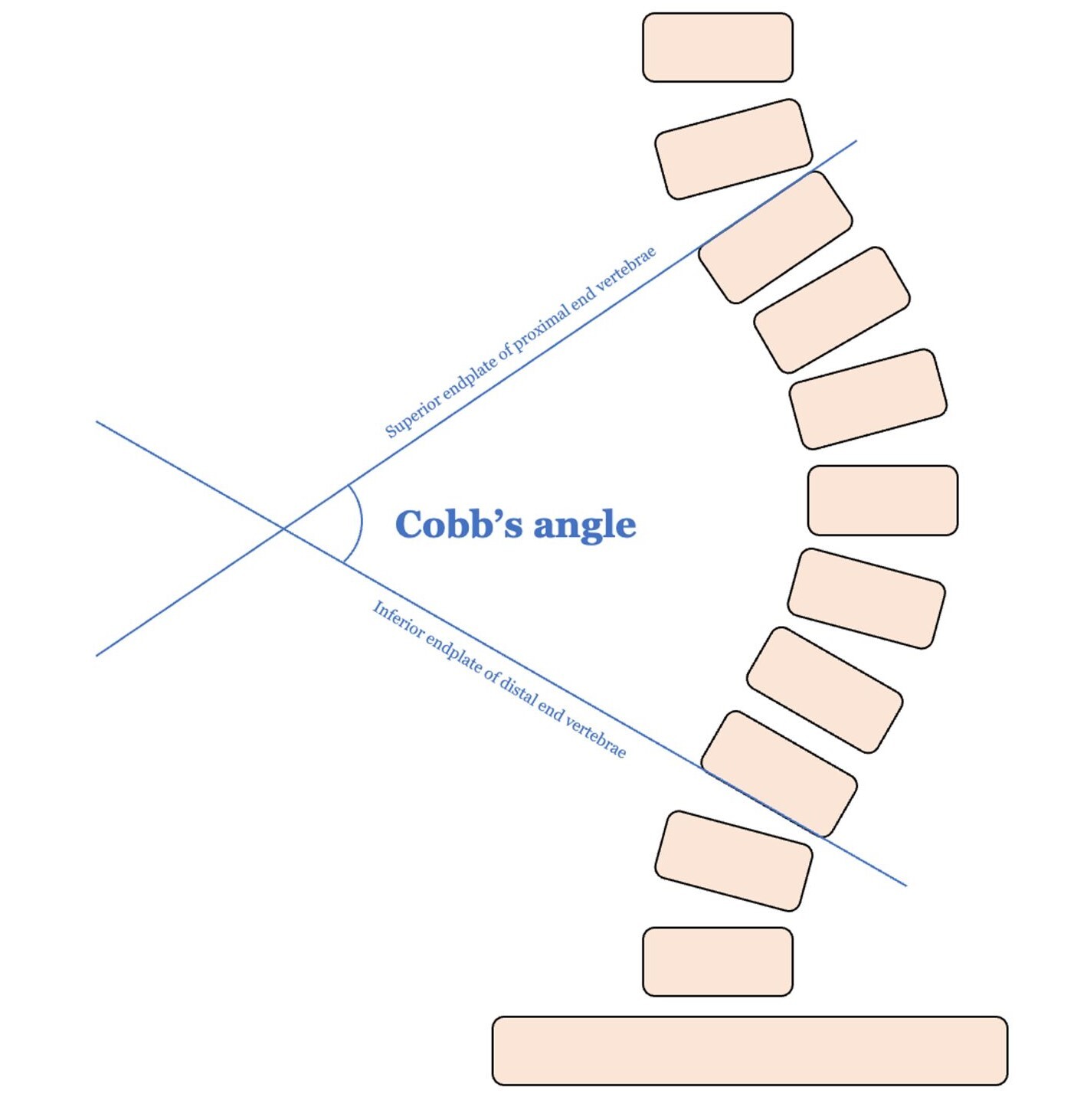
Cobb angle, defined by the angle between the endplates of the most tilted vertebrae at the curve’s apex, remains the gold standard for assessing scoliosis severity (Figure 1). However, its measurement has major drawbacks: it is time-consuming, prone to human error, and subject to significant inter- and intra-observer variability of up to 10º difference between observers. Determining which vertebrae to use for measurement can be especially challenging in cases with multiple curves (Jin et al. 2022; Kai Chen et al. 2021; Tu et al. 2019; Ha, Do, Bartret, et al. 2022; Sun et al. 2022). At the institutional level in Singapore, where over 120,000 children are screened yearly, this translates into over 14,000 scoliosis radiographs and approximately 1,500 physician hours annually, costing more than SGD$200,000 (~ USD$150,000) in labour (H.-K. Wong et al. 2005).
Automating Cobb angle measurement offers several benefits: improved accuracy and reproducibility, faster workflows, scalability, and the ability to decentralize scoliosis detection to non-specialists. While still a relatively nascent field, advances in deep learning and computer vision suggest automation of Cobb angle is now feasible. (Kai Chen et al. 2021; Tu et al. 2019; Ha, Do, Bartret, et al. 2022; Sun et al. 2022). Together with computer scientists and engineers, our medical team co-developed a novel software algorithm utilizing to automate Cobb angle measurement using computer vision.
This study aimed to evaluate the accuracy of a computer vision algorithm in measuring Cobb angle compared to manual measurement by fellowship-trained orthopaedic surgeons. Second, we sought to compare the speed of measurement between algorithm and manual measurements and evaluate the impact based on manpower time and cost savings based on utilizing computer vision.
Methods
Approval was obtained from our tertiary children’s hospital’s institutional review board. We developed a computer vision-based algorithm to automate the process of measuring Cobb angle measurement from DICOM-format plain coronal scoliosis radiographs. The model was trained using a dataset of 500 images manually annotated by two fellowship-trained paediatric orthopaedic surgeons. The application is run locally with a model built on Tensorflow Keras using Python, while the frontend and backend were developed on React and Flask accordingly. The software is a computer vision model utilising YOLOV5 for object detection, as well as Unet and EfficientNetB4 for segmentation. Performance characteristics include 267 neural network layers, 46,108,278 trainable parameters and a computational load of 107.6 Giga Floating-point Operations Per Second (GFLOPS) per inference. With 90.8% precision and 93.8% recall, the system meets clinical accuracy requirements while providing fast, reliable processing.
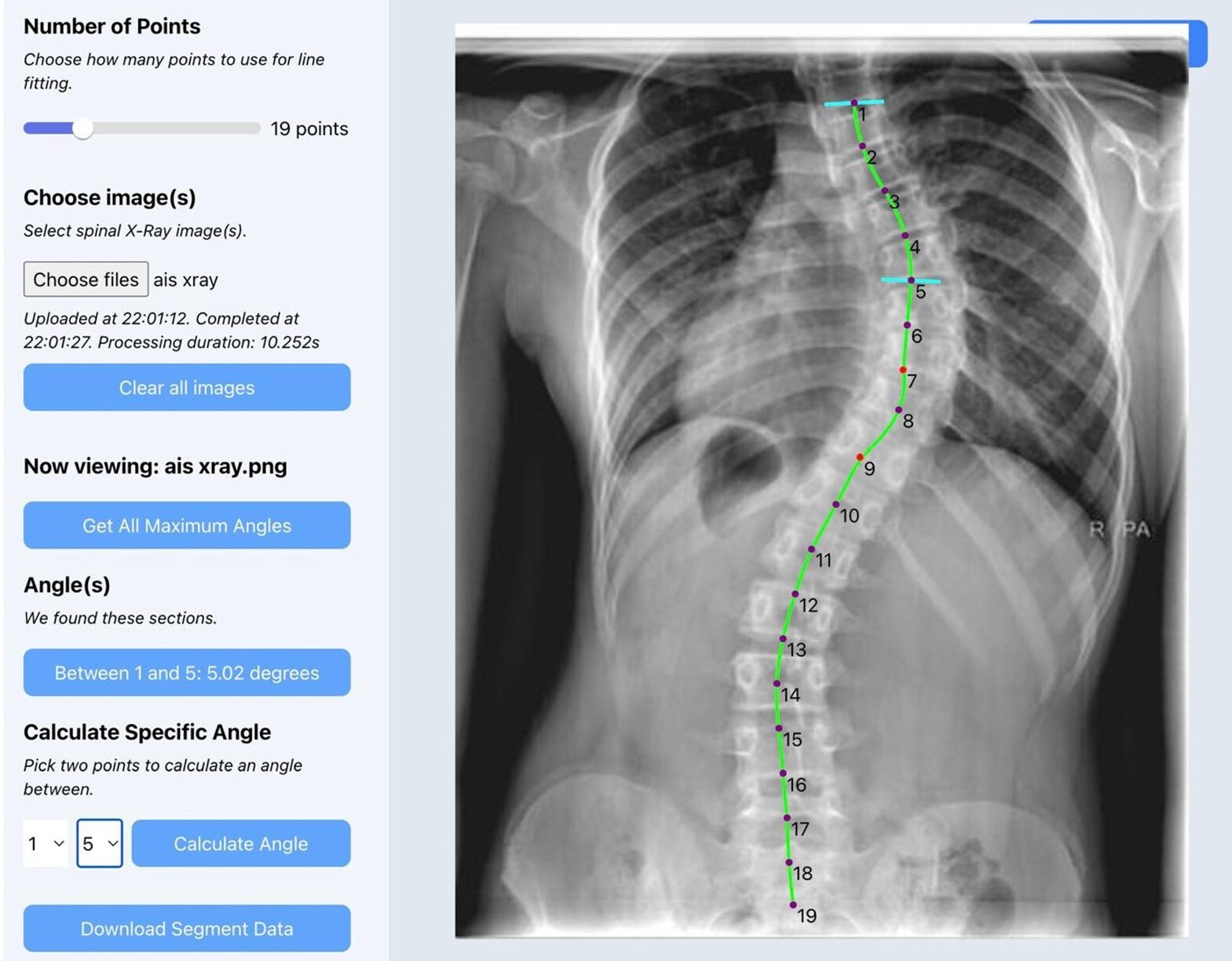
After uploading a radiograph, the software performs spinal segmentation, identifies vertebral endplates, and calculates the Cobb angle(s). For patients with more than one spinal curve, the software detects and displays the largest angles automatically. Users are given the option to refine the measurement manually or choose among alternate curves. Once finalized, users can export the radiograph with annotated angles as a PNG file, and all measurement data can be saved for further analysis. No patients were involved in the design or conduct of this study.
Algorithm performance
The algorithm’s performance was validated using a separate testing set of 54 radiographs with a Cobb angle of at least 10 o. Two orthopaedic surgery senior residents independently measure each radiograph, and the algorithm’s results were compared against the average of their measurements. Three primary metrics were analysed:
-
Maximum Cobb angle (in º)
-
Presence of multiple curves (yes/no)
-
Time to measurement (in seconds)
Additionally, orthopaedic surgery residents who regularly run paediatric scoliosis clinics used the software and provided qualitative feedback on usability and integration potential.
Results
Initial performance testing on a set of 54 plain coronal scoliosis radiographs with a range of Cobb angles from 10.4 to 59.7o shows that the algorithm can correctly identify scoliosis in 100% of films and correctly segment multiple curves within the same spine. The software measured a maximum Cobb angle with a mean angle difference of 4.50 + 5.19o compared to the manual measurement, which is within range of up to 10o inter-observer variability between physician-to-physician (Ha, Do, Bartret, et al. 2022; Sun et al. 2022). Mean age of the patients in the training set was 13.8 years, with 70.9% comprising of females.
The mean time taken from image upload to Cobb angle output was 9.76s, with >95% of cases completed under 10 seconds. In contrast, manual measurement by physicians averaged around 3 minutes per radiograph. This represented a time savings of over 90%.
Figure 2 demonstrates the software user interface accurately measuring Cobb angles on a plain coronal scoliosis radiograph.
Qualitatively, all of 13 orthopaedic surgery physicians found the interface intuitive and helpful, noting that the automated recommendations made the clinic workflow more efficient. Important features they highlighted included the ability to make fine adjustments to the measurements, as well as the ability to further annotate the radiographs, export the image, and integrate with existing pictures archiving and communications systems (PACS).
Preliminary cost-effectiveness analyses suggest that cost of the AI software is the most influential on the overall costs. To be cost-saving, extra cost of the software must be offset by manpower costs incurred, with a threshold of SGD$15 (or about USD$11) per AI analysis identified to be cost-effective based on our clinical workload.
Discussion
This study demonstrates that computer vision is a viable approach to automating Cobb angle measurement in adolescent idiopathic scoliosis. The algorithm achieved a mean measurement difference of 4.5º compared to orthopaedic surgery senior residents, with a time savings of over 90%. This is well within the clinically accepted threshold of 5º between AI and manual measurements (Li, Qian, Yan, et al. 2024; J. C. Wong et al. 2023; Wang et al. 2024). Preliminary feedback from potential end users has also been promising, with interest in clinical implementation to improve clinic efficiency and reduce patient wait times.
Accurate Cobb angle measurement remains technically challenging due to variability in identifying end vertebrae, localizing endplates, and delineating vertebral boundaries, all of which are affected by image quality, deformity severity, and observer experience (Jin et al. 2022; Langensiepen, Semler, Sobottke, et al. 2013). Artificial intelligence models face similar difficulties, requiring large, well-annotated datasets to generalize across diverse radiographs and maintain accuracy in severe deformities or poor image contrast. Unlike other radiologic AI applications that rely on broader pattern recognition, scoliosis assessment demands precise localization of multiple small anatomical landmarks, making high precision and robustness essential for reliable Cobb angle estimation (Kenneth Chen et al. 2024; Meng, Zhu, Yang, et al. 2025; Rajpurkar and Lungren 2023).
The current version of our application is deployed on a standalone laptop in our specialist scoliosis clinic separate from the existing electronic health records and is undergoing clinical trials to determine inter-observer kappa and mean angle difference to determine performance in comparison to the existing physician measurement. Concurrently, ongoing training of the algorithm with annotated scoliosis radiographs and software refinement are being carried out to improve algorithm performance and usefulness to users. Additionally, outcomes such as total time spent in the clinic by patients and physicians, clinic waiting time, direct and indirect costs of consultation, clinical outcomes, and patient and physician satisfaction are being measured for cost-effectiveness analyses.
In the future, we plan for integration with existing picture archiving and communications systems to allow for saving of annotations and measurement within electronic health records and cloud deployment. Additional adjacent markets and potential use cases for the software algorithm include identification of spinal curvature angles in adult spinal deformity and spinopelvic dissociation, in pre-operative templating and planning, and intra-operative radiographs for assessment of adequacy of deformity correction. Expansion to sagittal radiographs represents another key step, as accurate evaluation of sagittal parameters such as kyphosis and lordosis is integral to comprehensive spinal deformity assessment.
The limitations of our current study includes a small sample size of users and limited number of radiographs for both training and testing. A full clinical trial and cost-effectiveness analysis assessing the performance of the software algorithm in the clinical setting are required to fully determine the potential benefits of our solution.
Conclusion
This technical study shows that the computer vision software algorithm can reliably and efficiently measure multiple Cobb angles in a plain coronal scoliosis radiograph in less than 10 seconds. The software algorithm can potentially be deployed to improve accuracy and significantly reduce manpower hours required for manual Cobb angle measurements, a major benefit in high-volume orthopaedic clinics and nationwide screening programs. Additionally, it has potential uses in pre-operative surgical planning and intra-operative alignment checks.