Introduction
Anterior cruciate ligament (ACL) injuries are some of the most common and devastating injuries among athletes. There are estimated to be 250,000 ACL injuries per year with most of these injuries being suffered by athletes who are in their late teens to early 20s (Sutton and Bullock 2013). Additionally, ACL injuries are two to eight times more common in the female population than in the male population (Sutton and Bullock 2013; Kaeding, Léger-St-Jean, and Magnussen 2017). A substantial proportion of these injuries require ACL reconstruction (ACLR) which requires additional recovery time and has inherent limitations (Grindem et al. 2016). The graft harvest morbidity rate after ACLR averages to nearly 47%, which includes difficulty kneeling and anterior knee pain, while long-term studies have demonstrated elevated rates of reinjury and post-traumatic osteoarthritis decades after reconstruction (Selin et al. 2025; Grassi et al. 2022; Kunze et al. 2023).
Given the complications that arise from traditional ACLR, the Bridge-Enhanced ACL Restoration (BEAR) technique is emerging as an alternative surgical method for addressing ACL injuries. Unlike ACLR, which removes the torn ACL remnant tissue and replaces it with a graft, the BEAR technique preserves the ACL remnant tissue and connects them via a collagen and extracellular matrix scaffold (McMillan et al. 2022; Cleveland Clinic, n.d.). This BEAR implant is hydrated with the patient’s blood before implantation into the knee to encourage blood clot formation, which allows the ACL to heal without interruption from synovial fluid (McMillan et al. 2022; “BEAR Implant | Healthcare Professionals,” n.d.). While in the knee, the implant and native ACL tissue promotes cell proliferation and migration, allowing for ACL regeneration and revascularization (Grassi et al. 2022). The biologically driven repair process is mediated through a series of molecular and cellular mechanisms initiated by the implant’s collagen matrix. Bovine type 1 collagen functions as a provisional extracellular scaffold that facilitates platelet degranulation and the localized release of growth factors, including PDGF and TGF-β, which promote fibroblast migration and tenocyte proliferation at the repair site (Murray, Spindler, Devin, et al. 2006). Over time, the scaffold is gradually resorbed and replaced by native ligament tissue, resulting in a biological structure that closely resembles the native ACL.
Indications for BEAR candidacy depend heavily on the nature of the ACL tear and include these main criteria: the tear is midsubstance or proximal, and a substantial portion of the torn ACL is attached to the tibia (Shah et al. 2025). A recent change was made for the on-label indication for the BEAR procedure. The age indication has now been updated to include patients two years and older and is now an on label indication for partial tears of the ACL. Evidence suggests that individuals with open growth plates often do not experience growth disturbances after ACL surgery, enabling pediatric patients to be considered for BEAR as well (Gicquel, Geffroy, Robert, et al. 2018). Given that the BEAR avoids graft comorbidities, preserves the native ACL structure and produces similar functional outcomes to ACLR, this novel technique can be well-suited for young and active patients (Murray, Kalish, Fleming, et al. 2019; Shah et al. 2025). This case report documents a long-term patient outcome after undergoing the BEAR.
Overview and Injury Mechanism
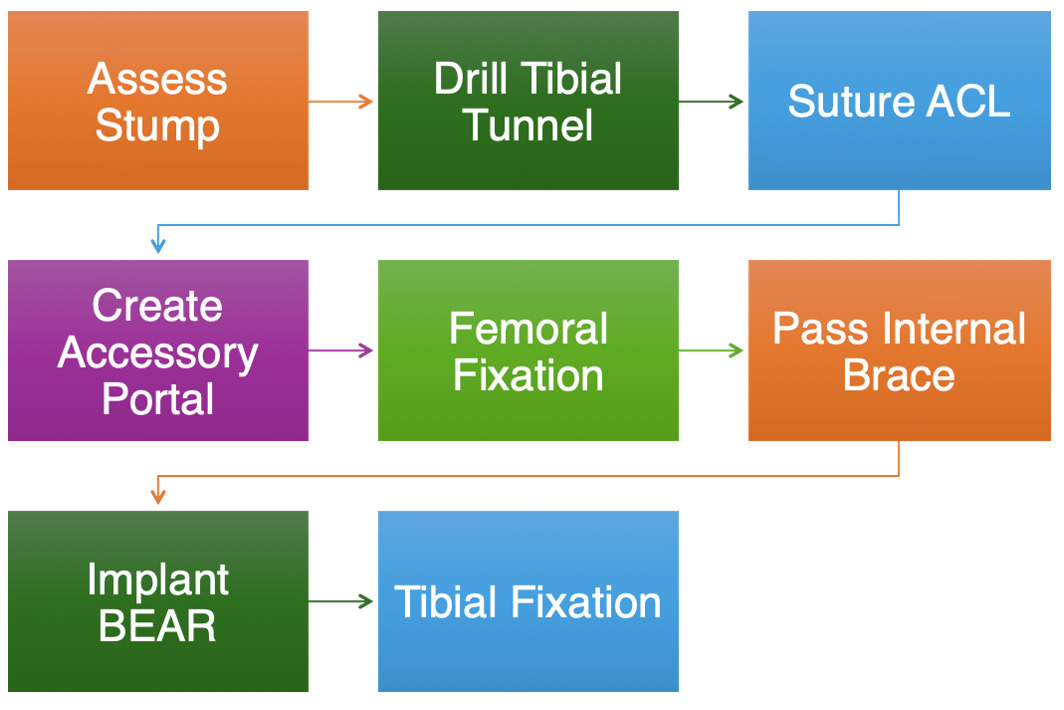
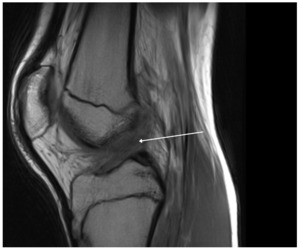
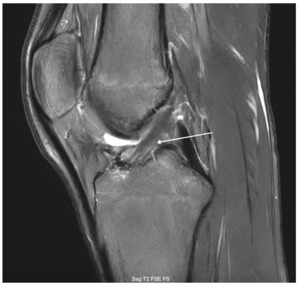
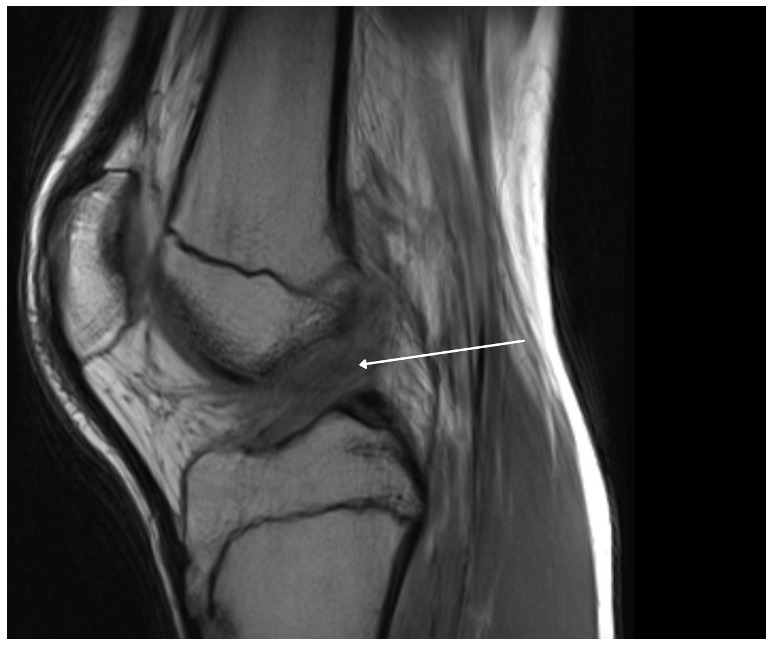
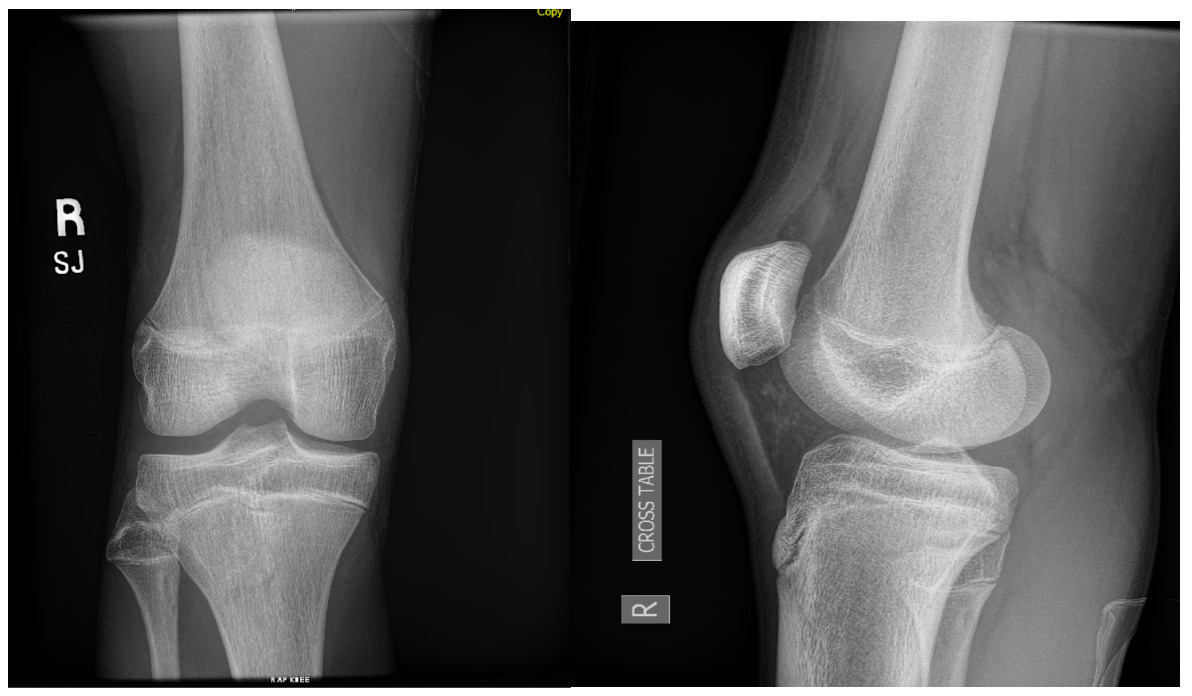
A 15-year-old male weighing 150 pounds and standing 6 ft 1 in tall, with no past medical history, presented to the clinic with buckling, catching and locking of the medial knee. He is a high school athlete who sustained a non-contact injury involving knee flexion combined with external rotation during a football game. He immediately reported a pop in the knee followed by stiffness and swelling. Clinical examination revealed joint effusion and medial and lateral joint line tenderness, on the inferior pole of the patella and tibial tubercle. Passive range of motion was limited, and both Lachman and McMurray tests were positive. Radiographs confirmed open physes with no evidence of fracture or malalignment., MRI demonstrated bony contusions and a substantial ligament injury, with a high-grade partial ACL tear, along with questions of peripheral posterior medial meniscus root tearing (Figures 1, 2). Management options were discussed with the patient, and he elected to proceed with surgical fixation. An additional consent was obtained from parental guidance for the possible publication of the surgical technique.
Surgical Technique
Surgical intervention was performed 6 weeks after the initial injury. Examination under anesthesia confirmed a positive Lachman test, grade 2+, without a definite endpoint.. Portals were created on the knee, and the joint was insufflated with fluid for diagnostic arthroscopy. The patellofemoral joint was in excellent condition, with no indication of medial or lateral meniscus tear or chondromalacia. At this point, the ACL stump was recognized and preserved, and the over-the-top guidance was used to locate the ACL’s origin on the femur. A Beath pin was then easily inserted, and a suture was used for later internal brace suture passage. The tibial tunnel was then created just anterior to the tibial stump. A fibrous anteriorly flipped ACL tissue remnant was identified at this point, and the stump was whipstitched using a suture passing device, and the cyclops was incorporated into the repair. The button was then brought onto the table, and the stumps sutures were carried up and through the button before passing through the lateral femoral cortex. An open arthrotomy was subsequently created to facilitate ACL repair and restoration. At this point, the sutures from the femoral button were passed down and through the BEAR implant, and 20 cubic centimeters (cc) of whole blood was splayed over the implant (Video 1a-b). Sutures from the implant were then passed down the tibial tunnel, and the BEAR implant was digitally inserted through a larger open medial arthrotomy incision for ACL repair and restoration. After positioning the BEAR, the knee was extended fully, and sutures through the BEAR implant were inserted into another button through the open tibial incision. Sutures were then tied down over the tibial button, and 3-0 Monocryl was used to close incisions. The sequence steps of the BEAR surgical technique are summarized in the flow chart (Figure 5)
Outcome
Postoperatively, the patient was placed in a postoperative hinged brace with a fixed range of motion of 0 to 90 degrees. Crutch protocol was instituted, and the patient remained non-weight bearing for the first 2 weeks. At follow-up, the patient showed primary healing at all incisions, an appropriate range of motion (0-90°), and continued current brace settings and was advanced to 50% partial weight bearing for the following 2 ½ weeks. Strengthening exercises with physical therapy began 2 weeks post-operatively per the recommended postoperative BEAR physical therapy protocol, focusing on pain and swelling. The following four phases consisted of closed chain strengthening, low impact cardio, plyometrics, and return to sport functional testing over a 6-8 month period (Shah et al. 2025).
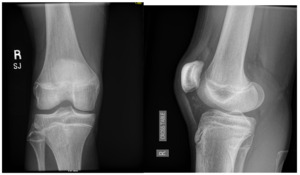
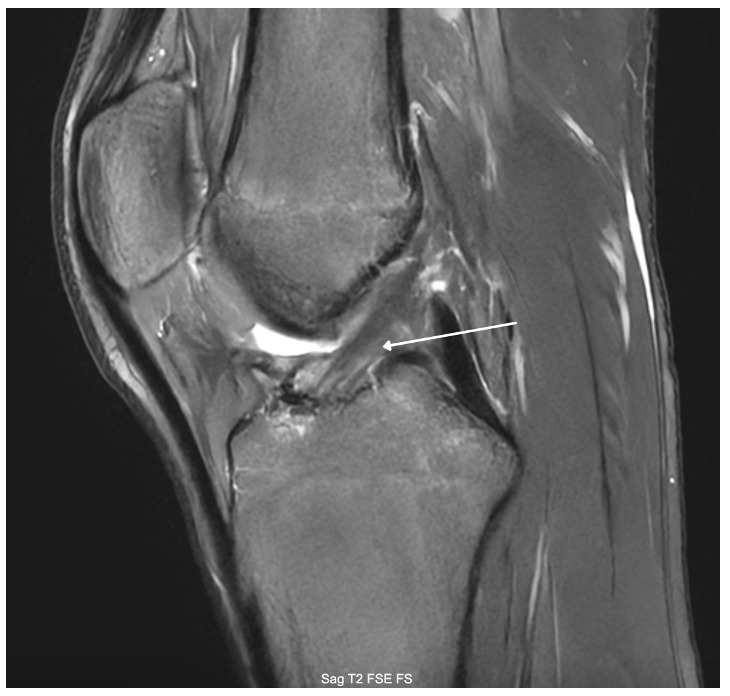
At the 6-month-follow-up, the patient reported no pain at the surgical sites and had regained full range of motion and strength of the hamstring and quadriceps (5/5). He also had a full range of motion. At 7-month-post surgery, an MRI was obtained to evaluate graft maturation prior to return to sport. The scan demonstrated an intact ACL with a robust graft, consisting of biologic healing (Figure 3). The patient was advised to continue aggressive strengthening over the next 4 weeks in anticipation of the upcoming football season. He was cleared for return to sport at approximately 8 months postoperatively. At the 1-year follow-up, the patient has done exceptionally well with no significant issues or complaints and was able to complete his football season, and was preparing for basketball season. He then revisited 18 months postoperatively where he demonstrated full knee flexion and extension (Figure 4a-b) and received the Lysholm Knee Score (LKS), which increased from 67/100 to 100/100, and a Knee Injury and Osteoarthritis Outcome Score (KOOS) with a base of 61/100, which increased to 94/100.
Discussion
This case report highlights the novel application of bridged-enhanced ACL restoration (BEAR) to improve outcomes following ACL injuries. Unlike conventional ACL reconstruction, which requires tendon grafting, BEAR employs a resorbable implant that facilitates healing of the torn ACL by utilizing the body’s natural repair mechanisms (Bond et al. 2024; Kantrowitz et al. 2024; Dhammi, null, and Kumar 2015). This technique eliminates complications associated with graft harvesting, such as donor site morbidity and postoperative graft failure (Barnett, Murray, Badger, et al. 2021). Additionally, BEAR circumvents common pitfalls of traditional reconstruction, including tunnel-related complications (Chau and Pandya 2025). Meta-analyses comparing BEAR to graft-based ACL reconstruction have demonstrated superior improvements in hamstring strength, faster symptom resolution, and earlier return to activities, along with higher ACL Return to Sports after injury scores (ACL-RTS) 6 months postoperatively (Mansour et al. 2023).
The BEAR has matured from early feasibility to randomized and post-market evidence. In a Level-1 RCT of mainly young adolescents with closed physes, BEAR demonstrated non-inferiorly to ACLR at 2 years. Contemporary reviews aggregating BEAR I-III trials report a combined retear rate of 9 of 64 (14%) of patients for BEAR and 2 of 35 (6.0%) for ACLR, supporting BEAR as a viable alternative to ACLR in select indications as real-world evidence continues to grow (Murray, Fleming, Badger, et al. 2020). Consistent with these findings, our patient achieved full range of motion and strength of the hamstring and quadriceps at 6-month follow-up, without surgical site pain. At 1-year, the patient successfully returned to competitive football, underscoring the technique’s capacity for effective rehabilitation while avoiding graft-related complications.
In younger athletic populations undergoing revision ACL reconstruction, there is a higher incidence of meniscus and cartilage abnormalities, graft failures, and poorer functional outcomes.(Ouillette et al. 2019) When treating adolescents and pediatric patients, special considerations must be given to their immature skeletons and open growth plates, which necessitate minimizing the drilling of tunnels in the tibia and femur, a step commonly associated with traditional ACL reconstruction techniques (Alharby 2010). Pediatric ACLR literature indicates that growth-disturbance risk is minimized when tunnels are small, central, and fully filled (McConkey, Bonasia, and Amendola 2011). The BEAR implant’s current instructions for use recommend a modified technique in patients to ensure adequate epiphyseal-sparing to accommodate a 2.4 mm drill pin with 2-3 mm bone margins to preserve the physis. More recently, the use of bioinductive anchors has become routine, replacing cortical buttons, further minimizing the risk of growth plate disturbance. (“BEAR Implant | Healthcare Professionals,” n.d.) Because the implant is positioned intra-articularly, it offers a growth-plate-sparing alternative that avoids limb-length discrepancy or angular deformities. By prioritizing growth plate preservation and native ligament healing, the BEAR procedure can be utilized to better support long-term joint integrity, reduce the risk of complications, and improve overall functional outcomes in the adolescent athletic and general population.
Our patient’s active lifestyle and age were important factors in selecting an appropriate clinical intervention, making him a strong candidate for the BEAR procedure. The decision was guided by his skeletal immaturity and high athletic demand, aligning with current literature that supports BEAR as a favorable option for young athletes aiming to minimize physeal disturbance and avoid graft-related complications. At 18-month follow-up, the patient demonstrated full recovery of quadriceps and hamstring functionality, with marked improvements in LKS and KOOS scores, corroborated by postoperative physical exam findings and MRI imaging. We will continue to structure surveillance through the skeletal maturity of the patient, including clinical exams (stability, ROM), growth assessment (leg-length) and imaging only if indicated. There will be surveillance MRI reserved for any recurrence of symptoms or for periodic evaluation of graft integrity as the patient continues to mature.
While this case highlights the successful recovery of an adolescent athlete following the BEAR procedure, it represents a single patient experience and therefore limits its generalizability. Additionally, due to the relative novelty of the technique and its limited use in clinical practice, long-term comparative data beyond two years remain sparse, particularly in younger populations. Careful patient selection remains an important consideration, as the procedure may be less suited for patients with poor tissue quality or experience chronic tears. Despite these limitations, this case contributes to the growing body of literature supporting BEAR as a viable biologic alternative to traditional ACL reconstruction, especially in adolescents. Future studies with longer follow-up periods and larger cohort sizes are essential to better assess the effectiveness, durability, and safety of BEAR across broader clinical scenarios.
Conclusion
This study demonstrates the successful long-term outcomes of a patient undergoing the bridged-enhanced ACL restoration (BEAR) procedure. This case highlights the trends towards repairs instead of reconstructions in ACL injuries for candidates that meet the requirements, particularly young athletes and individuals with occupational or recreational demands, and those with an intact tibial stump. The positive outcomes in this case support the advantages of biologic augmentation over traditional reconstruction methods. However, long-term follow-up studies are needed to fully assess and validate the efficacy, longevity, and benefits of this novel technique.
Human ethics
Consent was waived or obtained by all participants in this study

._summary_of_the_preparation_and_fixing_sequence_for_the_bear_implant._the_internal.png)
._18-months_postoperatively__the_patient_demonstrated_full_restoration_of_knee_ran.png)
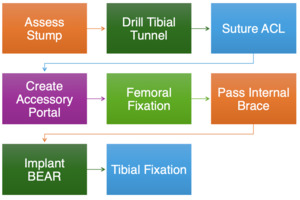
._summary_of_the_preparation_and_fixing_sequence_for_the_bear_implant._the_internal.png)
._18-months_postoperatively__the_patient_demonstrated_full_restoration_of_knee_ran.png)